Are all neutrons identical
Review: Neutrons are all identical to each other, just as protons are. Atoms of a particular element must have the same number of protons but can have different numbers of neutrons.
Are all electrons identical?
Every electron in the universe has exactly the same mass, exactly the same charge, and if you think about it, there’s no reason why they’d have to be. … According to him, the reason that every electron is the same is because every electron is the same electron.
Is the same number of neutrons?
ElementC: Carbon, no specific isotopeIsotope/Nuclide14 C : Carbon-14 specifically.
Is a neutron equal?
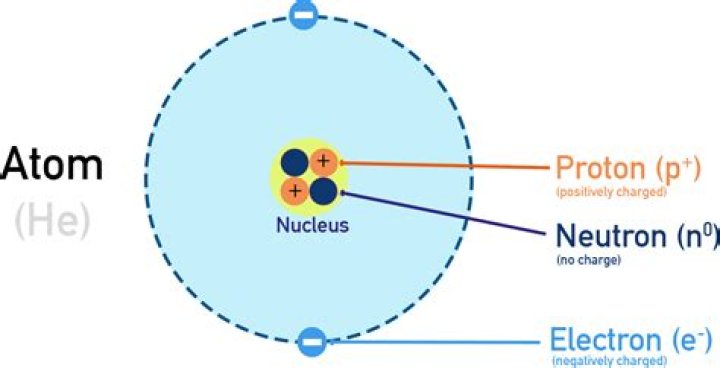
The number of protons in the nucleus of the atom is equal to the atomic number (Z). … The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).Are all atoms identical?
each element is made up of identical particles of matter called atoms. all the atoms in a pure substance or element are identical to one another but different to the atoms in a different element.
Is there only one photon?
Photons really only exist at the interactions of EM and material. In between EM is a wave. Hence the wave particle duality of light. However, from a point of view of Feynman diagrams you could be right.
Why are all protons identical?
They’re identical because they’re excitations of the same quantum field.
Why do neutrons exist?
Neutrons are required for the stability of nuclei, with the exception of the single-proton hydrogen nucleus. Neutrons are produced copiously in nuclear fission and fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes.Do all neutrons have the same mass?
Like protons, neutrons are bound into the atom’s nucleus as a result of the strong nuclear force. Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron).
How does an electron differ from a neutron?Answer : The electron differ from a neutron because, electron has relative charge of -1 u whereas neutron has no relative charge. Also, electron has relative mass of 1/1840 u and neutron has relative mass of 1 u.
Article first time published onWhy are protons not equal neutrons?
It might be a coincidence if they are same but protons should be equal to electrons…. Electrons are equal to protons so that positive and negative charges are balanced and the atom is electrically neutral…. Since neutrons have no charge it doesn’t matter them being equal to protons…..
Which elements have the same amount of neutrons?
Chemical SymbolNamePbLeadPlumbum
Are the neutrons and protons equal?
For most of the 16 lightest elements (up to oxygen) the number of neutrons is equal to the number of protons. … Its atomic number is 14 and its atomic mass is 28. The most common isotope of uranium has 92 protons and 146 neutrons. Its atomic number is 92 and its atomic mass is 238 (92 + 146).
Why are all atoms identical?
– Atoms of a given element are identical (meaning they have the same mass and properties). – Atoms retain their identity in all chemical reactions. This is basically the Law of Conservation of matter stating that an equal amount of mass exists before and after a reaction.
Can atoms be destroyed?
No atoms are destroyed or created. The bottom line is: Matter cycles through the universe in many different forms. In any physical or chemical change, matter doesn’t appear or disappear. Atoms created in the stars (a very, very long time ago) make up every living and nonliving thing on Earth—even you.
Are atoms unbreakable?
Atoms are not indivisible or indestructible. … While all atoms of a given element do have the same number of protons which gives them a great deal of their chemical character, they can vary in their mass and properties by changes in their number of neutrons and electrons.
Do all protons look alike?
All protons are identical. For example, hydrogen protons are exactly the same as protons of helium and all other elements, or pure substances. However, atoms of different elements have different numbers of protons. … The number of protons in an atom determines the electrical charge of the nucleus.
What makes a quark?
A quark is an elementary particle which makes up hadrons, the most stable of which are protons and neutrons. Atoms are made of protons, neutrons and electrons. … Neutrons and protons are made up of quarks, which are held together by gluons. There are six types of quarks.
Do all protons have the same mass?
To answer your specific question: protons are stable, so they all have the same mass.
What is light made of?
Light is made of particles called photons, bundles of the electromagnetic field that carry a specific amount of energy.
Is light a wave or particle?
Light Is Also a Particle! Now that the dual nature of light as “both a particle and a wave” has been proved, its essential theory was further evolved from electromagnetics into quantum mechanics. Einstein believed light is a particle (photon) and the flow of photons is a wave.
Does light have a thickness?
Because we assume the light as a point particle and the point haven’t any size. We can’t tell the width or thickness of point particle. Example – 1 – If you determine light behaves like a particle then we can say that it has no any type of size.
Is neutron heavier than proton?
The neutron is very slightly heavier than the proton, by about 0.1%, or 1.00137841887 according to the best measurements. … The neutron, as it happens, has a little more mass (and thus energy) than a proton and an electron combined.
Does a neutron have a mass of 1?
Atomic mass units (amu) are useful, because, as you can see, the mass of a proton and the mass of a neutron are almost exactly 1 in this unit system.
Did Chadwick discover the neutron?
In 1932, Chadwick made a fundamental discovery in the domain of nuclear science: he proved the existence of neutrons – elementary particles devoid of any electrical charge. … For this epoch-making discovery he was awarded the Hughes Medal of the Royal Society in 1932, and subsequently the Nobel Prize for Physics in 1935.
Are neutrons useless?
A neutron, by itself, is not stable. It is slightly heavier than a proton, and, left alone, decays into a proton+electron after a few minutes (and an antineutrino). Within a stable nucleus, neutrons cannot decay because there is no room for an extra proton with the quantum state that would result from such a decay.
Is a neutron positive or negative?
Among atomic particles, the neutron seems the most aptly named: Unlike the positively charged proton or the negatively charged electron, neutrons have a charge of zero.
Are neutrons a positive charge?
Proton—positive; electron—negative; neutron—no charge. The charge on the proton and electron are exactly the same size but opposite. The same number of protons and electrons exactly cancel one another in a neutral atom.
What is the difference between proton neutron and electron?
The difference between electron, proton and neutron is the charge they carry. Electrons are charged negative, protons are charged negative, and neutrons do not carry any charge. … An electron’s atomic mass unit is (5.45×10–4). The orbits of electrons are located outside the nucleus of the atoms.
How does an electron differ from an atom?
Electrons are the negatively charged particles of atom. Together, all of the electrons of an atom create a negative charge that balances the positive charge of the protons in the atomic nucleus. Electrons are extremely small compared to all of the other parts of the atom.
How many electrons can the fourth energy level hold?
Regardless of its shape, each orbital can hold a maximum of two electrons. Energy level I has just one orbital, so two electrons will fill this energy level. Energy level IV has 16 orbitals, so it takes 32 maximum electrons to fill this energy level.