Can Solvolysis be e1
Adding a weak base to the reaction disfavors E2, essentially pushing towards the E1 pathway. In many instances, solvolysis occurs rather than using a base to deprotonate. This means heat is added to the solution, and the solvent itself deprotonates a hydrogen. The medium can effect the pathway of the reaction as well.
Does solvolysis happen in E1?
Adding a weak base to the reaction disfavors E2, essentially pushing towards the E1 pathway. In many instances, solvolysis occurs rather than using a base to deprotonate. This means heat is added to the solution, and the solvent itself deprotonates a hydrogen. The medium can effect the pathway of the reaction as well.
What determines solvolysis?
solvolysis, a chemical reaction in which the solvent, such as water or alcohol, is one of the reagents and is present in great excess of that required for the reaction. … The solvents act as or produce electron-rich atoms or groups of atoms (nucleophiles) that displace an atom or group in the substrate molecule.
Is solvolysis SN1 or E1?
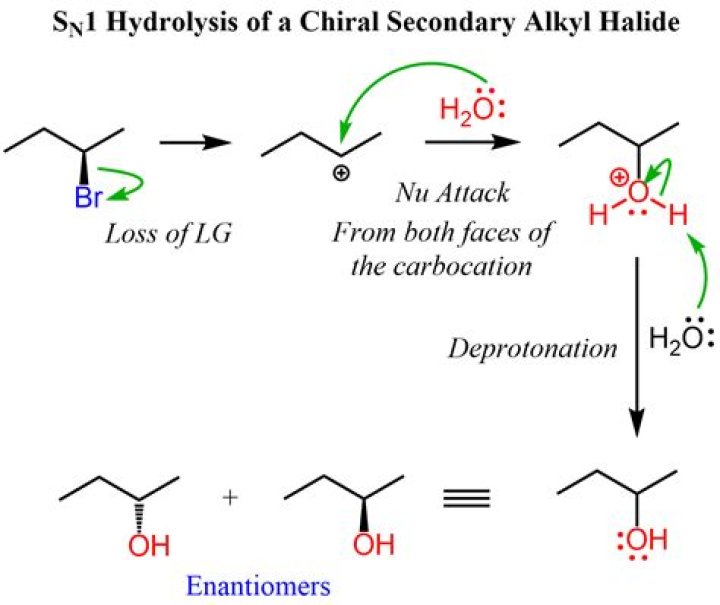
Solvolysis is a type of nucleophilic substitution (SN1/SN2) or elimination where the nucleophile is a solvent molecule. Characteristic of SN1 reactions, solvolysis of a chiral reactant affords the racemate.What is the difference between solvolysis and hydrolysis?
is that hydrolysis is (chemistry) a chemical process of decomposition involving the splitting of a bond and the addition of the hydrogen cation and the hydroxide anion of water while solvolysis is (chemistry) any reaction, between a solute and its solvent, in which one or more bonds of the solute are broken; …
Can SN2 be solvolysis?
In SN2 solvolysis reactions, the nucleophile is involved in the rate-determining process. As a result, stronger nucleophiles react more quickly.
Does E1 prefer tertiary?
The rate of SN2 reactions goes primary > secondary > tertiary. The “big barrier” to the SN1 and E1 reactions is carbocation stability. The rate of SN1 and E1 reactions proceeds in the order tertiary > secondary > primary.
What undergoes solvolysis in methanol most rapidly?
Which one of the following alkylbromides undergoes most rapid solvolysis in methanol solution to give corresponding methyl ether ? Tertiary alkyl undergoes solvolysis in either acetic acid or in ethanol.What is solvolysis in organic chemistry?
Solvolysis: A reaction in which the solvent is a reactant, and becomes part of the reaction product. Hydrolysis of tert-butyl chloride; solvent = water.
What is solvolysis used for?Solvolysis Process refers to a thermochemical process leading to depolymerization of UP composites, using hot water as a reactant.
Article first time published onIn which solvent is solvolysis faster?
The more polar the solvent, the better it is at solvating the developing carbocation and lowering the activation energy. An 80:20 water/acetone mixture is more polar than a 70:30 mixture, so the solvolysis is faster in the more water-rich solvent.
Which solvent is best for solvolysis of alkyl halide?
What about the SN1? The most useful application of SN1 reactions in synthesis is in “solvolysis” reactions, where the alkyl halide is dissolved in a nucleophilic solvent such as water or an alcohol. This works best for tertiary alkyl halides.
Why does Solvolysis favor SN1?
This reaction occurs via SN1 because Cl- is a good leaving group and the solvent is polar protic. This is an example of a solvolysis reaction because the nucleophile is also the solvent.
How do you do an E1 reaction?
Putting It Together: The E1 Mechanism Proceeds Through Loss Of A Leaving Group, Then Deprotonation. The reaction is proposed to occur in two steps: first, the leaving group leaves, forming a carbocation. Second, base removes a proton, forming the alkene.
Is oh a good leaving group?
Alcohols have hydroxyl groups (OH) which are not good leaving groups. … Because good leaving groups are weak bases, and the hydroxide ion (HO–) is a strong base.
Which salt is commonly used to increase the rate of solvolysis of alkyl halides?
2>1>3.
Is methanol volatile?
Methanol appears as a colorless fairly volatile liquid with a faintly sweet pungent odor like that of ethyl alcohol. Completely mixes with water. … It is an alkyl alcohol, a one-carbon compound, a volatile organic compound and a primary alcohol. It is a conjugate acid of a methoxide.
Is hydrolysis a substitution?
Hydrolysis is a special type of nucleophilic substitution (SN1) where water acts as both nucleophile and a solvent molecule.
Is E1cB Unimolecular?
The name of the mechanism – E1cB – stands for Elimination Unimolecular conjugate Base. Elimination refers to the fact that the mechanism is an elimination reaction and will lose two substituents. Unimolecular refers to the fact that the rate-determining step of this reaction only involves one molecular entity.
Is etoh a strong base?
Still, ethanol has the ability to act as an acid because of the ability to donate it’s hydroxyl proton. However, aqueous solutions of ethanol are slightly basic. This is is because the oxygen in ethanol has lone electron pairs capable of accepting protons, and thus ethanol can act as a weak base.
How do you know if it's E1 or E2?
The key differences between the E2 and E1 mechanism are: 1) E2 is a concerted mechanism where all the bonds are broken and formed in a single step. The E1, on the other hand, is a stepwise mechanism. … 3) E2 is a second-order reaction and the rate depends on the concentration of both, the substrate and the base.
Which chloro hydrocarbons readily undergo solvolysis?
Solvolysis is related to stability of carbocation formed and among these benzyl carbocation is most stable. So, it will undergo solvolysis readily.
Is MeOH a strong nucleophile?
Methanol lacks stabilizing effects, so although it can act as a base, it is fairly weak one. It is, however, a pretty decent nucleophile given its small size and lone pairs.
Which one undergoes solvolysis in acetic acid at the fastest rate?
tert-Butyl chloride undergoes solvolysis in both acetic acid and formic acid. Solvolysis occurs 5000 times faster in one of these two solvents than in the other.
Which bromide will most rapidly undergo solvolysis in aqueous ethanol?
h. tert-Butyl chloride undergoes SN1 solvolysis in aqueous ethanol more rapidly than does tert-butyl bromide under the same conditions.
How many transition states are involved in the solvolysis reaction?
We show that all three transition states, in both protic and in aprotic solvents, are highly dipolar and are strong hydrogen bond acids and strong hydrogen bond bases, except for the tert-butyl iodide transition state in aprotic solvents, which has a rather low hydrogen bond acidity.
Which reacts by e1 mechanism in ethanol most readily?
Ghere ethanol reacts as a nucleophile attacking the carbocation generated by the heterolysis of the halide. Since p-methoxybenzyl bromide yields the most stable carbocation, it reacts most readily with ethanol.
Which will undergo fastest SN2?
1-chloro-2-methyl-hexane undergoes the fastest under SN2 because it’s alkyl halide is a primary alkyl halide which is favored by SN2.
Which compound is most nucleophilic?
In both laboratory and biological organic chemistry, the most common nucleophilic atoms are oxygen, nitrogen, and sulfur, and the most common nucleophilic compounds and functional groups are water/hydroxide ion, alcohols, phenols, amines, thiols, and sometimes carboxylates.
Which of the following is tertiary halide?
CH3Cl.
What makes a leaving group good?
Good leaving groups are weak bases. They’re happy and stable on their own. Some examples of weak bases: halide ions (I-, Br-, Cl-) water (OH2), and sulfonates such as p-toluenesulfonate (OTs) and methanesulfonate (OMs). The weaker the base, the better the leaving group.

