Do peptide bonds form spontaneously
Then, peptide bond formation occurs spontaneously at the surface of water, facilitated by the formation of the copper complex at the interface.
Why peptide bond formation is not spontaneous?
Based on a quite old reference 1 (which I’m using because it’s available free by open access), peptide bond formation at 25 C is unfavorable only because of a large enthalpy change, on the order of 1.5 kcal/mol (6.3 kJ/mol).
Is the formation of a peptide bond Exergonic?
The formation of the peptide bond is an endergonic reaction that requires energy, which is obtained from ATP in living beings. Because this reaction involves the removal of a water molecule, it is called a dehydration synthesis reaction.
How are peptide bonds formed?
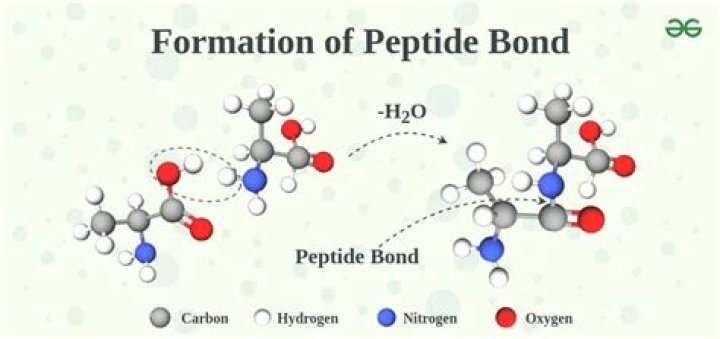
A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other molecule, releasing a molecule of water (H2O). This is a dehydration synthesis reaction (also known as a condensation reaction), and usually occurs between amino acids.Does peptide bond formation require energy?
The formation of the peptide bond consumes energy, which, in organisms, is derived from ATP. Peptides and proteins are chains of amino acids held together by peptide bonds (and sometimes by a few isopeptide bonds).
Why is the peptide bond which is thermodynamically unstable resistant to spontaneous hydrolysis?
3) Why is the peptide bond, which is thermodynamically unstable, resistant to spontaneous hydrolysis? A peptide bond is stabilized by resonance, which gives the carbonyl-carbon-to-amide-nitrogen link partial double-bond character.
Where does the energy for peptide bond formation come from?
The formation of the peptide bond is catalyzed by peptidyl transferase, an RNA-based enzyme that is integrated into the large ribosomal subunit. The energy for the peptide bond formation is derived from GTP hydrolysis, which is catalyzed by a separate elongation factor.
How is a protein formed from amino acids?
Proteins are formed in a condensation reaction when amino acid molecules join together and a water molecule is removed. The new bond formed in protein molecules where amino acids have joined (-CONH) is called an amide link or a peptide link.How is peptide bond formed in translation?
During translation, peptide bonds are formed from the amino (N) to the carboxyl (C) terminus by removal of water (also referred to as dehydration or condensation) and catalyzed by RNA (referred to as a ribozyme) that forms part of the ribosome. … This bond is fixed in one plane.
How are peptides formed from amino acids?Within a protein, multiple amino acids are linked together by peptide bonds, thereby forming a long chain. Peptide bonds are formed by a biochemical reaction that extracts a water molecule as it joins the amino group of one amino acid to the carboxyl group of a neighboring amino acid.
Article first time published onHow is a peptide bond formed between two amino acids to form a dipeptide?
A dipeptide is formed when two Amino acids join together by one Peptide bond. This happens via a Condensation Reaction. The bond between the two amino acids forms between the carboxyl group on one and the amino group on another, therefore producing a water molecule as a product.
Is formation of a peptide bond endothermic or exothermic?
You made a bond between the two amino acids. That is exothermic.
Are peptide bonds hydrogen bonds?
Hydrogen bonding between atoms in peptide bonds is a common theme in protein structure and forms the basis for all secondary structure. Figure 1 Amide hydrogen atoms carry a partial-positive charge. The nitrogen of the peptide bond carries a substantial amount of positive charge due to resonance.
How is a dipeptide formed quizlet?
How is a dipeptide formed? Condensation synthesis of two amino acids.
Why are peptide bonds stable?
The stability of the peptide bond is due to the resonance of amides. With resonance, the nitrogen is able to donate its lone pair of electrons to the carbonyl carbon and push electrons from the carbonyl double bond towards the oxygen, forming the oxygen anion.
Are peptide bonds kinetically stable?
This process is called a condensation reaction. One perhaps surprising fact about peptide bonds is that they are very kinetically stable, in fact, a peptide bond in a solution will last 1000 years if no catalyst is present to disrupt it. Many amino acids can be connected via peptide bonds, making a polypeptide.
Are peptide bonds covalent?
Covalent bonds involve the equal sharing of an electron pair by two atoms. Examples of important covalent bonds are peptide (amide) and disulfide bonds between amino acids, and C–C, C–O, and C–N bonds within amino acids.
Is peptide bond hydrolysis favorable?
Actually, in the first one or two weeks in biochemistry I was taught that peptide bond hydrolysis is thermodynamically favorable but kinetically unfavorable (half-life: about 1000 years). In the amount of time it would take to hydrolyze a peptide bond, another one would probably be formed in its place.
Are peptide bonds formed in transcription?
The basic building blocks of proteins. The process following transcription during which the nucleotide sequence of mRNA is read and ‘translated’ into a chain of amino acids (protein). …
Where does peptide bond formation occur in a bacterial ribosome and how?
Between the two amino acids (found on charged tRNA), bound to the two sites of the large sub units of bacterial ribosomes, when two charged tRNAs are brought close enough, peptide bond is formed with the help of ribozyme.
Does DNA form peptide bonds?
Peptide bonds are formed between the carboxylic acid group of one amino acid and the amino group of a second amino acid. … Nucleotide monomers linked via phosphodiester bonds make up nucleic acids (i.e., DNA and RNA).
Which amino acids can form ionic bonds?
Alanine Type: NonpolarArginine Type: IonicCysteine Type: PolarGlutamic Acid Type: IonicGlutamine Type: PolarGlycine Type: NonpolarHistidine Type: IonicIsoleucine Type: NonpolarLeucine Type: NonpolarLysine Type: Ionic
Do free amino acids have peptide bonds?
Free amino acids lack peptide bonds because they function as monomers that multiply and link via peptide bonds to form a protein chain. A free amino acid has two groups: a carbonyl group (- COOH) and a -NH2 group. … As a result, free amino acids lack a peptide chain.
Which amino acids can form hydrogen bonds?
The amino acids asparagine and glutamine posses amide groups in their side chains which are usually hydrogen-bonded whenever they occur in the interior of a protein.
Which bond is a peptide bond quizlet?
What is a peptide bond? The covalent bond (C-N) formed by a condensation reaction between two amino acids; links the residues in peptides and proteins.
How do amino acids join a peptide bond?
The bond that holds together the two amino acids is a peptide bond, or a covalent chemical bond between two compounds (in this case, two amino acids). It occurs when the carboxylic group of one molecule reacts with the amino group of the other molecule, linking the two molecules and releasing a water molecule.
How peptide bonds are formed how many peptide bonds are present in a Tetrapeptide?
Peptide bond is formed by the union of a-amino acids with the loss of water molecules. Three peptide bonds are present in a tetra-peptide.
When an ionic bond forms electrons are?
Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. The atom that loses the electrons becomes a positively charged ion (cation), while the one that gains them becomes a negatively charged ion (anion).
Are peptide and amide bonds the same?
Amide and peptide bonds are biochemical bonds. The key difference between amide and peptide bond is that an amide bond forms between a hydroxyl group and an amino group of two molecules whereas a peptide bond forms between two amino acid molecules during the formation of a peptide chain.
Are peptide bonds polar?
Polar covalent bond is a covalent bond in which the atoms have an unequal attraction for electrons and so the sharing is unequal. Hence the peptide bond is a nonpolar covalent bond because it holds together two amino acids. Hence the peptide bond is nonpolar.
What type of reaction occurs spontaneously?
The answer is E, exergonic. Exergonic reactions are characterized by the potential energy of their reactants and products.