How does pH affect peroxidase activity
The activity of the peroxidase enzyme is dependent on pH. … Lower pH levels result in a more effective inactivation of the enzyme than do higher pH levels, meaning that acidic environments are more effective inhibitors of peroxidase activity than basic environments.
Which pH was ideal for peroxidase activity?
results showed that the pH 6.0 is the optimum pH for peroxidase activity, while the activity decreased in pH below and above 6.0.
How does pH level affect enzyme activity?
Enzymes are also sensitive to pH . Changing the pH of its surroundings will also change the shape of the active site of an enzyme. … This contributes to the folding of the enzyme molecule, its shape, and the shape of the active site. Changing the pH will affect the charges on the amino acid molecules.
What factors affect peroxidase activity?
The results of this experiment demonstrated that the performance of hydrogen peroxidase was greatly affected by all three factors, temperature, concentration level, and pH.What is the effect of temperature on peroxidase activity?
We can conclude that increase in temperature from 30 to 40 ºC change probably structure of peroxidase to ordered shape, so that activity of enzyme in 30 and 40 ºC increased and in more temperature (60 and 70 ºC) enzyme slowly denature and activity decreased.
How does pH affect the rate at which catalase breaks down hydrogen peroxide?
Catalase pH Levels In humans, catalase works only between pH 7 and pH 11. If the pH level is lower than 7 or higher than 11, the enzyme becomes denaturated and loses its structure. The liver sustains a neutral pH of about 7, which creates the best environment for catalase and other enzymes.
How does pH affect enzyme activity Google Scholar?
pH also plays a role in enzyme activity. For hydrogen peroxidase, the highest activity was at pH 7. At pH 11, the activity was very low and at pH 1 there was no activity. This is likely because pH affects ionic and hydrogen bonds which are important to enzyme shape and therefore enzyme activity (Reece, et al 2010).
What is the optimal pH for turnip peroxidase?
The partially purified turnip enzyme was stable at a pH range 2.6−6.0 and had an optimum pH at 4.0.What is the pH of the liver?
The liver maintains a neutral pH (about pH 7), which is easiest for its enzymes, such as catalase, to work in.
How does a low pH affect enzyme activity?At extremely low pH values, this interference causes the protein to unfold, the shape of the active site is no longer complementary to the substrate molecule and the reaction can no longer be catalysed by the enzyme. The enzyme has been denatured.
Article first time published onHow does a high pH affect an enzyme activity quizlet?
As pH increases, enzyme activity increases until it reaches an optimal point in which enzymes denatures and as pH increases, enzyme activity decreases. … They can also interact with the active site and disrupt hydrogen binding and binding to ionic residues decreasing, or in some cases increasing, activity of the enzyme.
How does pH affect salivary amylase activity?
Effect of pH The optimum pH for the enzymatic activity of salivary amylase ranges from 6 to 7. Above and below this range, the reaction rate reduces as enzymes get denaturated. The enzyme salivary amylase is most active at pH 6.8. … So the salivary amylase does not function once it enters the stomach.
How does pH affect polyphenol oxidase?
As shown in Fig. 2, polyphenol oxidase activity was pH-dependent, with complete inactivation at 2.0 ≤ pH > 10. The maximum PPO enzyme activity from buriti pulp observed at pH 7.0 was 14.69 U.mL−1, using catechol as substrate.
How does pH affect the rate of the enzymatic browning?
Higher acidity, such as a pH below 4.0, inhibits the activity of the polyphenol oxidase (PPO) enzyme. Adding citric, ascorbic or other acids, such as vinegar, lowers the pH and prevent enzymatic browning. … Cooking fruits or vegetables inactivates (destroys) the PPO enzyme, and enzymatic browning will no longer occur.
Why does peroxidase break down hydrogen peroxide?
Peroxidase is an enzyme found in a wide variety of organisms, from plants to humans to bacteria. Its function is to break down hydrogen peroxide (H2O2), which is one of the toxins produced as a byproduct of using oxygen for respiration. (The fact that it’s toxic is what makes hydrogen peroxide useful in first aid kits.
What factors affect enzyme activity lab?
- Temperature (use water baths to minimise fluctuations)
- pH (acidic or alkaline solutions)
- Substrate concentration (choose range to avoid saturation)
- Presence of inhibitor (type of inhibitor will be enzyme-specific)
What is enzyme optimum pH?
Optimum Enzyme pH Most enzymes’ optimum pH is neutral or close to neutral, like amylase found in saliva, which has an optimal pH = 6.8. Some enzymes prefer a more drastic pH, like pepsin, which can have an optimum pH of 1.7 to 2. Sometimes enzyme pH optima depends on where the enzyme is found.
How does pH affect the decomposition of hydrogen peroxide?
pH tests revealed that H2O2 was more stable at low pHs in that a 12% decrease in the concentration of H2O2 occurred at pH 11.5-12 over 3 h (Fig. … Mathre (1971) also reported that the increase in pH leads to high consumption of H2O2 increases particularly at >11 in the presence of cyanide. …
Why does pH affect catalase activity?
At extremely high pH levels, the charge of the enzyme will be altered. This changes protein solubility and overall shape. This change in shape of the active site diminishes its ability to bind to the substrate, thus annulling the function of the enzyme (catalase in this case).
How does pH affect the oxidation of catechol?
At pH 7, the enzyme is most active and rapidly catalyzes the oxidation of catechol to give the dark, brown color.
How does nephron regulate blood pH?
The kidneys have two main ways to maintain acid-base balance – their cells reabsorb bicarbonate HCO3− from the urine back to the blood and they secrete hydrogen H+ ions into the urine. By adjusting the amounts reabsorbed and secreted, they balance the bloodstream’s pH.
What does the pH indicate?
pH is a measure of how acidic/basic water is. The range goes from 0 – 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. pH is really a measure of the relative amount of free hydrogen and hydroxyl ions in the water. … pH is reported in “logarithmic units”.
At what pH is the rate of enzyme activity the lowest?
The lowest rate of enzyme activity is pH 4.
What is the role of peroxidase?
Peroxidases, also known as catalases, are also an oxidoreductase class of enzymes, which catalyze oxidoreduction reactions. The peroxidase enzyme catalyzes the decomposition of hydrogen peroxide into water and molecular oxygen (see illustration). Catalase is a haem-containing enzyme.
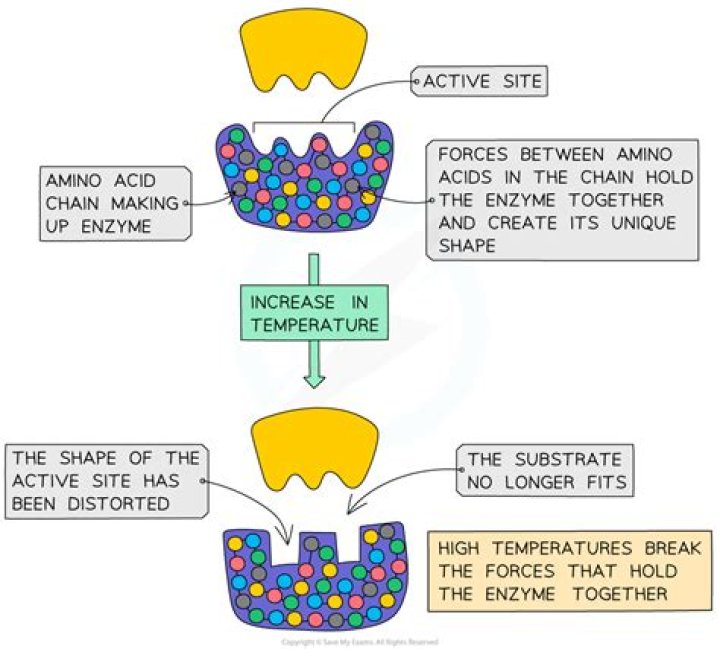
What happens if the pH is above or below the optimum pH?
This effect can be permanent and irreversible and is called denaturation. The diagram below shows what happens to an enzyme when denaturation occurs. Each enzyme has an optimum pH but it also has a working range of pH values at which it will still work well. This depends on the type of enzyme.
What happens to turnip peroxidase during and after the reaction?
What happens to turnip peroxidase during and after the reaction? During the reaction, the activation energy is lowered when the peroxidase is present. This results in a faster reaction time. … If you omitted parts of the reaction to the mix, the reaction would not react as it should.
What happens when the pH is 2?
A pH of 2 is 10 times more acidic than a pH of 3, and 100 times more acidic than a reading of 4.
How does pH affect ionic bonds?
The two that pH changes affect are salt bridges (a) and hydrogen bonding (b). Salt bridges are ionic bonds between positively and negatively charged side chains of amino acids. … Increasing the pH by adding a base converts the -NH+3 ion to a neutral -NH2 group.
What effect do changes in pH have on enzymes IB Biology?
Extreme pH levels will produce denaturation. The structure of the enzyme is changed. The active site is distorted and the substrate molecules will no longer fit in it. At pH values slightly different from the enzyme’s optimum value, small changes in the charges of the enzyme and its substrate molecules will occur.
What happens to the rate of reaction as the pH changes?
Optimal pH increases enzyme rate of reaction while less than optimal pH decreases it. Increasing temperature also increases enzyme rate of reaction, until things get too hot, then the enzyme denatures and ceases to function. Denaturing an enzyme essentially destroys it.
How does environmental pH affect reaction rate?
Environmental pH can alter the efficiency of enzyme activity, including through disruption of hydrogen bonds that provide enzyme structure. Each enzyme has an optimum pH range. … Once all of the substrate is bound, the reaction will no longer speed up, since there will be nothing for additional enzymes to bind to.

