How much has the ocean acidified
Ocean acidity has increased about 25% from preindustrial times to the early 21st century, a pace faster than any known in Earth’s geologic past. The acidity of the ocean is greater than any point in the past two million years.
How much has ocean level changed pH?
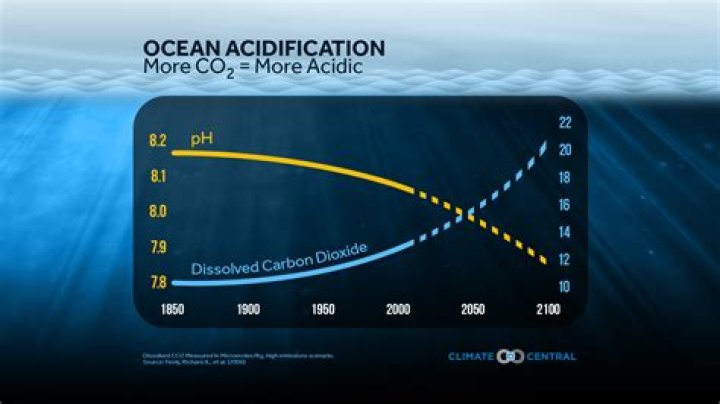
During this time, the pH of surface ocean waters has fallen by 0.1 pH units. This might not sound like much, but the pH scale is logarithmic, so this change represents approximately a 30 percent increase in acidity.
How much has ocean acidification increased since the 18th century?
Since industrialization began in the 18th century, surface-ocean acidity has increased by 30%.
What was the ocean pH in 2000?
The globally and annually-averaged surface ocean pHT2000 in the Atlantic, Pacific, and Indian Oceans (60°N to 60°S) is 8.07 ± 0.02 (1σ uncertainty on gridded values, see Methods), 8.06 ± 0.03, and 8.07 ± 0.02, respectively, with a global average of 8.07 ± 0.02 between 60°N to 60°S.Is ocean water slightly acidic?
Seawater is slightly basic (meaning pH > 7), and ocean acidification involves a shift towards pH-neutral conditions rather than a transition to acidic conditions (pH < 7).
What is ocean alkalinity?
Alkalinity is the excess of proton acceptors (bases) in a solution. … In the ocean, the bases of the carbon dioxide system (bicarbonate and carbonate) dominate alkalinity and oceanographers define alkalinity based on a titration of seawater with a strong acid.
What was the ocean's pH in 2008?
YearpH20058.07920068.07720078.07520088.073
What is the pH of milk?
Cow’s milk Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it’s acid-forming or alkaline-forming.What pH is soap?
Results: Majority of the soaps have a pH within the range of 9-10. Majority of the shampoos have a pH within the range of 6-7. Conclusions: The soaps and shampoos commonly used by the population at large have a pH outside the range of normal skin and hair pH values.
Is ocean acidification bad?For good reason, ocean acidification is called “osteoporosis of the sea.” Ocean acidification can create conditions that eat away at the minerals that oysters, clams, lobsters, shrimp, coral reefs, some seaweed plants and other marine life use to build their shells and skeletons. Human health, too, is a concern.
Article first time published onWhat does a drop in ocean pH from 8.2 to 8.1 mean?
The ocean is not acidic! Seawater is never going to get below pH 7—so you must not know what you’re talking about.” … The scale is not linear—a drop from pH 8.2 to 8.1 indicates a 30 percent increase in acidity, or concentration of hydrogen ions; a drop from 8.1 to 7.9 indicates a 150 percent increase in acidity.
What pH is acid rain?
The pH scale measures how acidic an object is. … Normal, clean rain has a pH value of between 5.0 and 5.5, which is slightly acidic. However, when rain combines with sulfur dioxide or nitrogen oxides—produced from power plants and automobiles—the rain becomes much more acidic. Typical acid rain has a pH value of 4.0.
What was causing the mussel shells to erode?
Habitat loss has been identified as the most important factor causing the decline of mussel species throughout North America. Dams, channelization, and drainage of river systems have resulted in the elimination of 30 to 60% of the original mussel fauna residing in the impacted areas.
Is the ocean polluted?
Marine debris is a persistent pollution problem that reaches throughout the entire ocean and Great Lakes. Our ocean and waterways are polluted with a wide variety of marine debris, ranging from tiny microplastics, smaller than 5 mm, to derelict fishing gear and abandoned vessels.
What does pH stand for?
pH may look like it belongs on the periodic table of elements, but it’s actually a unit of measurement. The abbreviation pH stands for potential hydrogen, and it tells us how much hydrogen is in liquids—and how active the hydrogen ion is.
Is ocean acidification real?
Ocean acidification is real. … Ocean acidification is causing changes in seawater chemistry, leading to conditions that are corrosive to organisms that use calcium carbonate to make shells, skeletons, and other important body parts, such as young oysters.
What pH is salt water?
The average pH for sea water is 8.2 but can range between 7.5 and 8.5 depending on the local conditions.
How much has the sea level risen in the past 100 years?
Over the past 100 years, global temperatures have risen about 1 degree C (1.8 degrees F), with sea level response to that warming totaling about 160 to 210 mm (with about half of that amount occurring since 1993), or about 6 to 8 inches.
What is the pH of black coffee?
With an average pH of 4.85 to 5.10, most coffees are considered rather acidic. While this doesn’t present a problem for most coffee lovers, the acidity can negatively affect certain health conditions in some people, such as acid reflux and IBS.
What increases ocean pH?
As the amount of carbon dioxide in the atmosphere rises, the oceans absorb a lot of it. In the ocean, carbon dioxide reacts with seawater to form carbonic acid. This causes the acidity of seawater to increase.
What happens if alkalinity is high?
If your alkalinity level becomes too high, it will become difficult to change the pH. You’ll know your pH is too high when your pool water is cloudy, there is scale build up on your pool walls and your chlorine is no longer successfully sanitizing your pool.
How do you increase pH in ocean water?
An increase in ocean alkalinity could be achieved by dissolving rocks and minerals either directly in the open ocean or through engineered systems. This would lead to a build-up of calcium, magnesium, or sodium ions in seawater and thus an uptake of CO2 to form bicarbonate ions.
What pH is a lemon?
Lemon juice has a pH between 2 and 3, which means it’s 10,000–100,000 times more acidic than water (1, 2, 3). A food’s pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.
What pH is toothpaste?
The pH of the toothpaste can generally range from 7 to 10, depending on its additives.
What pH is bleach?
Bleach: pH 11-13. Bleach is one of the most common cleaning supplies in households and commercial settings. This particular product has a pH between 11 and 13. Its high level of alkalinity is what makes it corrosive.
Is honey an acid?
According to the National Honey Board, the acidity of honey ranges from a pH of about 3.4 to about 6.1, with an average of 3.9. The acidity of any honey is directly related to the floral sources that created it.
What is the pH of Coke?
How acidic is Coke? Its pH is reported to be 2.6 to 2.7, mainly due to H3PO4, phosphoric acid. As a fizzy drink, it contains plenty of dissolved carbon dioxide, but this makes very little contribution to the acidity.
What is the pH of a egg?
The pH of a fresh egg yolk is about 6.0 and increases to 6.4 to 6.9 during storage. Storage at refrigerated temperatures greatly slows the pH change and helps reduce the rate of the thick egg white from thinning. In general, the egg pH is stable and does not disrupt food product formulations.
What role do zooplankton play in carbon cycle?
Within this food web, zooplankton serve both as trophic links between primary producers and higher trophic levels (such as fish) and as recyclers that transform particulate carbon and nutrients into dissolved pools.
What does acid do to marine shells?
Ocean acidification can negatively affect marine life, causing organisms’ shells and skeletons made from calcium carbonate to dissolve. The more acidic the ocean, the faster the shells dissolve.
What are scientists doing about ocean acidification?
EPA is collaborating with the National Estuary Program (NEP) and their partners to measure coastal acidification using autonomous, in situ monitoring sensors for dissolved carbon dioxide (pCO2) and pH in estuaries across the U.S. These state of the art sensors give scientists new insights into acidification in …

