Hydrogen Monochloride
How do you write the formula for hydrochloric acid?
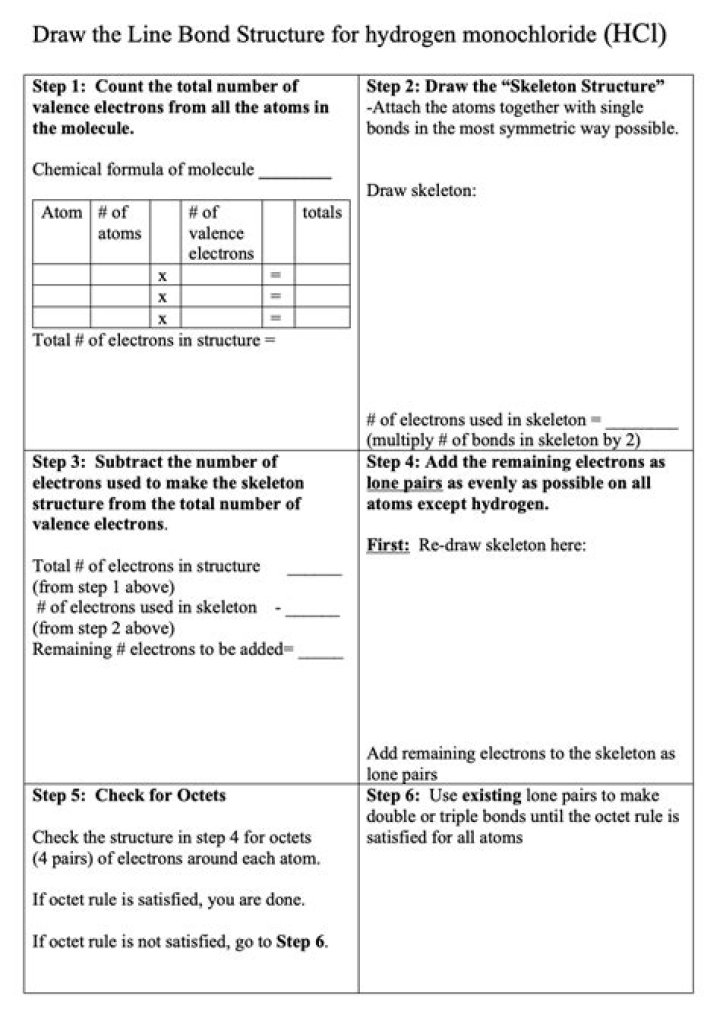
The hydrochloric acid compound has the chemical formula HCl and, as such, is a hydrogen halide.
Do you also know what the formula of hydrochloric acid is?
HClWhat is hydrochloric acid hydrochloric acid. Hydrochloric acid is a gas at room temperature. Solutions of hydrochloric acid in water are called hydrochloric acid. Hydrochloric acid is widely used in the chemical industry as a reagent in the production of other chemicals. Most are produced as a byproduct of reactions with chlorine.
How is hydrochloric acid made like this?
Hydrochloric acid can be formed from the direct combination of chlorine (Cl2) and hydrogen (H2) and the reaction is rapid at temperatures above 250 ° C (482 ° F). The reaction, represented by the equation H2 + Cl2 → 2HCl, is accompanied by heat dissipation and appears to be accelerated by humidity.
What does hydrochloric acid look like?
At room temperature, hydrochloric acid is a colorless to pale yellow, corrosive, non-flammable gas, heavier than air and emits a strongly irritating odor. In the air, hydrochloric acid forms dense, white and corrosive vapors. Hydrogen chloride can be released from volcanoes.
What is the difference between hydrochloric acid and hydrochloric acid?
Hydrochloric acid and hydrochloric acid have the same chemical formula: HCl. The difference between the two is that hydrochloric acid is a gas and hydrochloric acid is an aqueous solution. Hydrochloric acid is very reactive with metals.
What is the name of H2?
H2 is also called molecular hydrogen and is made up of two protons and two electrons. Hence, it is the most common form of hydrogen as it is stable with a neutral charge. H2 is not a free radical. It is the antioxidant of hydrogen-rich water. H2 is the smallest molecule in the universe.
What is the formula for Hydrogen Monochloride?
CHEBI: 17883 Hydrochloric acid The hydrochloric acid compound has the chemical formula HCl and is as such a hydrogen halide. At room temperature, a colorless gas is produced, which forms white vapors of hydrochloric acid in contact with atmospheric water vapor.
Is hydrochloric acid poisonous?
Portal of toxic substances Hydrochloric acid is a colorless, corrosive and non-flammable gas like vapors in air. Hydrochloric acid is not absorbed through the skin, but when hydrochloric acid gas comes into contact with moisture it forms hydrochloric acid, which is corrosive and can cause irritation and burns.
Is HCl polar or non-polar?
HCl is a polar molecule because chlorine has a higher electronegativity than hydrogen. It then attracts electrons to pass more time, which gives it a negative charge and hydrogen a positive charge.
How do you know if Br2 is polar or non-polar?
Where is the hydrochloric acid?
Stomach
What does HCI mean in science?
Computer Interaction
Can Hydrogen Chloride Kill You?
Severe lung damage can result in death. Depending on the concentration and duration of exposure, hydrochloric acid can cause functional and morphological damage to the respiratory tract (Darmer et al.
What is hydrochloric acid used for?
Hydrochloric acid can form when many plastics are burned . Upon contact with water, hydrochloric acid forms acid.
How to make hydrochloric acid at home?
First you put some salt in an alembic. You make them react with each other.
excess hydrochloric acid leaks from the top of the
What is the strongest acid in the world?
Carboranic acids may be the strongest acid in the world, antimony pentafluoric Vinegar and hydrochloric acid are So it works as shown in cup # 2. In cup # 4 Mixing vinegar and salt produces hydrochloric acid.This dissolves the copper compound Salt is added to the vinegar and can clean the ear. flammable - aqueous solution of hydrochloric acid, a gas acid. Reacts with sulphides, carbides, borides and phosphides, releasing toxic or flammable gases. Reacts with many metals (including aluminum, zinc, calcium, magnesium, iron, tin and all alkali metals) to produce flammable hydrogen gas.
What is the pH of HCl?
CH4 has a neutral pH of about 7. On the other hand, substances such as hydrochloric acid, HCl, are held together by polar ionic bonds and in water hydrogen decomposes liquid acid. HCl therefore has a very low pH and is a very strong acid.
Hydrogen Monochloride
![Hydrogen Chloride Cannon]()