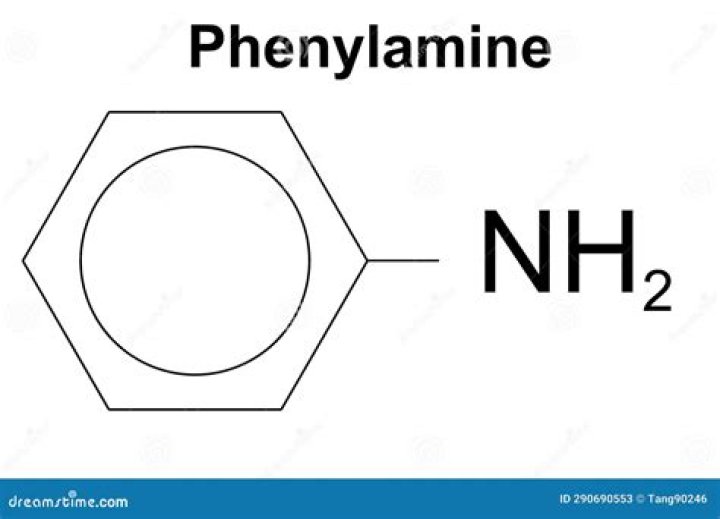
Is Phenylamine a base
Despite the fact that the phenylamine is only a very weak base, with a strong acid like hydrochloric acid the reaction is completely straightforward. Phenylamine is only very slightly soluble in water, but dissolves freely in dilute hydrochloric acid.
Is phenylamine a strong base?
Phenylamine has the structure: The lone pair on the nitrogen touches the delocalised ring electrons . . . … Taken together – the lack of intense charge around the nitrogen, and the need to break some delocalisation – this means that phenylamine is a very weak base indeed.
Is propylamine or phenylamine a stronger base?
The availability of a lone pair of electrons on a base determines its strength as it is these electrons that will “mop up” H+ ions in solution and hence increase pH towards more alkaline conditions. Therefore, phenylamine is a weaker base than propylamine because its lone pair is less available.
Are amines acids or bases?
Amine are basic and easily react with the hydrogen of acids which are electron poor as seen below. Amines are one of the only neutral functional groups which are considered basis which is a consequence of the presence of the lone pair electrons on the nitrogen.Is phenylamine a good Nucleophile?
Taken together – the lack of intense charge around the nitrogen, and the need to break some delocalisation – means that phenylamine is a very weak base indeed. These are reactions in which the phenylamine acts as a nucleophile.
Which is a stronger base ammonia or phenylamine?
Phenylamine is a weaker base than ammonia. The lone pair on the nitrogen of phenylamine is delocalised into the ring, thus making the lone pair less available to combine with hydrogen ions.
Why phenylamine is a weaker base?
The availability of a lone pair of electrons on a base determines its strength as it is these electrons that will “mop up” H+ ions in solution and hence increase pH towards more alkaline conditions. Therefore, phenylamine is a weaker base than ethylamine because its lone pair is less available.
Why are amine classified as bases?
Amines are also a base in the Lewis definition. An amine group has a lone pair of electrons when it forms three bonds. It can donate these electrons to other molecules, making it a base in the Lewis definition too.Do amines act as bases?
Amines are bases, and their basicity depends on the electronic properties of the substituents (alkyl groups enhance the basicity; aryl groups diminish it), steric hindrance, and the degree of solvation of the protonated amine.
Why are amines called base?According to the Lewis acid-base concept, amines can donate an electron pair, so they are Lewis bases. Also, Brønsted-Lowry bases can accept a proton to form substituted ammonium ions. So, amines are bases according to both the Lewis and the Brønsted-Lowry theories.
Article first time published onWhich amine is strongest base?
Amine Answers The amide ion is the strongest base since it has two pairs of non-bonding electrons (more electron-electron repulsion) compared to ammonia which only has one. Ammonium is not basic since it has no lone pair to donate as a base.
Why is butylamine stronger than ammonia?
Amines are weak bases. Alkylamines, e.g. ethylamine, are stronger bases than ammonia because the alkyl groups ‘push’ electrons towards the nitrogen atom, making it more electron- rich than the nitrogen in ammonia.
What makes a base stronger?
The less stable the molecule is (which happens when lots of electron density is concentrated on one atom), the more it will want to give some of its electron density by accepting a proton; therefore, the strongest base will be the one with the highest energy electrons (or the one with the most electron density …
Why is phenylamine insoluble in water but soluble in HCl?
Explain. Aniline does not undergo hydrogen bonding because of the presence of the benzene which is highly hydrophobic. therefore aniline is insoluble in water. In the HCl the amine group becomes protonated (-NH3+) and the ionic hence soluble in HCl.
Is phenylamine the same as Aminobenzene?
The key difference between phenylamine and aminobenzene is that the name phenylamine describes that aniline has a phenyl group and an amine group whereas the name aminobenzene describes that aniline has an amino group substituted to a benzene ring. … Therefore, these are two names for the same compound.
Is amino benzene basic?
Basicity. Aniline is a weak base. Aromatic amines such as aniline are, in general, much weaker bases than aliphatic amines.
Why is phenylamine insoluble in water?
Aniline is insoluble in water because of the large hydrocarbon part that retards the formation of an H-bond. So it is insoluble in water. Anilines are the organic compounds in the class of group coming in organic chemistry which are also called aminobenzene or phenylamine.
Is net3 a strong base?
This means the nitrile is the least basic of all of these molecules. The highest pKaH value here is 10.8 for triethylamine. That makes triethylamine the strongest base out of all of those listed.
Is Phenylamine more basic than amide?
The lone pair on nitrogen interacts with the delocalised pi system of benzene. Therefore it is less available for donation which makes phenylamine a weaker base than NH3. But phenylamine is still able to function as a base, hence it is more basic and has a lower pKb than amide.
Which is a stronger base ammonia or cyclohexylamine?
Cyclohexylamine is thus more basic than ammonia because alkyl groups are electron-donating (through the effect of hyperconjugation). The cyclohexyl group makes the N atom more nucleophilic than in ammonia, and hence more basic.
Which is more basic benzylamine or ammonia?
Factually, pkb value for Ammonia Is 4.75 whereas for benzylamine the pkb value is 4.70. Therefore, we can say that Benzylamine is more Basic…
Is ammonium chloride a base?
Ammonium chloride, a water-soluble salt of ammonia, is slightly acidic as a water-based solution. The acidic component of ammonium chloride (Cl-) produces hydrogen (H+) ions when dissolved in water. The basic component (NH4+) produces hydroxide (OH-) ions when dissolved in water.
Is an amide a base?
Compared to amines, amides are very weak bases and do not have clearly defined acid–base properties in water. On the other hand, amides are much stronger bases than esters, aldehydes, and ketones.
Is ammonia a base?
Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. … A weak base is one which doesn’t convert fully into hydroxide ions in solution.
What is basicity of amine?
Amines, like ammonia, are weak bases (K b = 10 −4 to 10 −6). This basicity is due to the unshared electron pair on the nitrogen atom.
Is NH an amine?
Primary amines have one carbon bonded to the nitrogen. … For amines, we are counting the carbons bonded to the nitrogen. Since nitrogen has a normal valence of three, we can also conclude that there are two N-H bonds in primary amines and one N-H bond in secondary amines. In tertiary amines there are no N-H bonds.
Why does NH2 act as a base?
Water is neutral whereas ammonia is a weak base (even less acidic). Stronger conjugate acids have weaker conjugate base and vice versa . Therefore, NH2- is a stronger base.
Is thiol a base?
Thiols are easily deprotonated. Relative to the alcohols, thiols are more acidic. The conjugate base of a thiol is called a thiolate.
Are tertiary amines Basic?
In the gas phase, amines exhibit the basicities predicted from the electron-releasing effects of the organic substituents. Thus tertiary amines are more basic than secondary amines, which are more basic than primary amines, and finally ammonia is least basic.
Why are amines weak bases?
Amines, unless they have four R- groups attached (a quaternary amine) have a lone pair of electrons, which just like the lone pair in ammonia, can accept a proton. Hence amines, like ammonia, are weak bases.
What amines are most basic?
Correspondingly, primary, secondary, and tertiary alkyl amines are more basic than ammonia.