Is there any effect of impurities on surface tension
Yes, by adding impurities the surface tension of water increases or decreases is determined by the cohesion between the molecules or the adhesion among the water molecules and impurity. … We can say that by adding oil to water, the surface tension of water decreases.
Does impurities affect surface tension?
As the concentration of the impurity increases the equilibrium surface tension decreases.
What is effect of impurities and temperature on surface tension?
(1) Effect of temperature : The surface tension of a liquid decreases with increasing temperature of the liquid. Hence, the angle of contact also decreases with increasing temperature of the liquid. (2) Effect of impurity : (1) The surface tension of a liquid increases with dissolved impurities like common salt.
What is the effect of impurity on the surface tension of liquid?
The presence of impurities either on the liquid surface or dissolved in it, considerably affect the force of surface tension, depeneding upon the degree of contamination. A highly soluble substance like sodium chloride when dissolved in water, increases the surface tension of water.Which factors affect the surface tension?
- The presence of any contamination or impurities.
- The presence of dissolved substances.
- Electrification.
- Temperature.
How do you increase surface tension?
To increase the surface tension of water, you’d have to add something to water, in which case, it would no longer be pure water. By adding something more polar than water to water, the surface tension would increase (just as doing the opposite decreases surface tension).
Why do soluble impurities increase surface tension?
As surface tension increases, intermolecular forces increase. … The Presence of ImpuritiesThe presence of impurities on the surface of, or dissolved in, a substance directly affects the surface tension of the liquid. The surface tension of water, for example, will increase when highly soluble impurities are added to it.
What is the effect of surface tension of liquid when sparingly soluble impurity added to it Shaalaa?
When / sparingly soluble substances are added surface tension decreases.What are the effect of impurity?
Impurities may bring about incompatibility with other substances. Impurities may lower the shelf life of the substances. Impurities may cause difficulties during formulations and use of the substances. Sometimes Impurities changes the physical and chemical properties of the substances.
What is the effect of impurity on angle of contact?Hence, angle of contact will increase. In case of impurities, when a highly soluble impurity like NaCl is added to water, surface tension increases whereas it decreases for sparingly solube (eg: phenol) or insoluble impurities. Hence, angle of contact (on addition of impurities) will decrease or increase accordingly.
Article first time published onWhat is the effect on surface tension of temperature?
Surface tension decreases with an increase in temperature. So these are the effects of temperature on viscosity and surface tension. In general, surface tension decreases when the temperature increases because cohesive forces decrease with increased molecular thermal activity.
What is the effect on surface tension of molten copper?
Answer: As the temperature increases, viscosity decreases. Surface tension decreases with increase in the temperature.
What are the effect of surface tension on angle of contact?
Surface tension generally decreases on increasing the temperature i.e adhesion increases. Hence, angle of contact will increase.
Does surface tension depend on surface area?
No, surface tension doesn’t depend on the surface area. … The surface tension primarily depends on the forces of attraction between the particles within the giving liquid and also on the gas, solid or liquid in contact with it.
Does surface area affect surface tension?
In liquids, when the surface area is increased, molecules are allowed to move from the bulk phase to the interface. As a result, the number of molecules per unit area (i.e. the distance between the molecules at the interface) remains the same. Therefore, the surface tension does not change.
How do you decrease surface tension?
Some liquids such as oil and kerosene can destroy surface tension in water. Adding soap or detergent reduces surface tension in water. Increasing the temperature of the liquid reduces surface tension.
Does gravity affect surface tension?
How does gravity affect surface tension? Answer 1: While changes in gravity would not affect the interactions within a liquid, gravitational forces along with surface tension determine the shape of liquid droplets in air or on various surfaces.
Does surface tension increase with intermolecular forces?
Surface tension is the energy required to increase the surface area of a liquid by a given amount. The stronger the intermolecular interactions, the greater the surface tension.
Does density affect surface tension?
After noting the density of each liquid it became clear that the higher the density the greater the surface tension. The experiment showed that water, the liquid with the highest density, has the highest surface tension; while alcohol,the liquid with the lowest density, has the lowest surface tension.
Does adding salt to water increase surface tension?
The surface tension of water is increased when salt is added to it. Although the strong interactions between sodium cations and partial negative oxygen, and chloride anions and partial positive hydrogens disrupt some hydrogen bonding between water molecules, they actually strengthen the surface tension of water.
Which has highest surface tension?
Water has the highest surface tension among the given liquids due to the presence of an inter-molecular hydrogen bonding.
Does sugar increase surface tension?
It has been reported in the literature that sugars such as dextrose and sucrose increase the surface tension of water. The effect was interpreted as a depletion of the solute molecules from the water-air interface.
Do impurities negatively impact the properties of substances?
Impurities can affect the physical and chemical properties of substances, resulting in inefficient or unwanted chemical reactions.
What is the effect of impurity on melting point?
The presence of even a small amount of impurity will lower a compound’s melting point by a few degrees and broaden the melting point temperature range. Because the impurity causes defects in the crystalline lattice, it is easier to overcome the intermolecular interactions between the molecules.
What is the effect of impurities on freezing point?
When an impurity is added its freezing point is lowered i.e. its freezing point decreases. The depression in freezing point increases with the increase in concentration of the solute because on adding the solute the vapour pressure of solution becomes lower than that of pure solvent.
How does impurity affect viscosity?
For the same reasons, we claim that fluid impurities can affect fluid mobility, as impurities will shift the phase diagram towards lower fluid density and viscosity, leading to stronger buoyancy forces and higher mobility.
When sparingly soluble insoluble substance is into water surface tension of water?
Hence, when a sparingly soluble substance like alcohol is dissolved in water, the surface tension of the water decreases. So, the correct answer is “Option B”.
What is an impact on surface tension when I impurity is increased II temperature is decreased?
In general, surface tension decreases when temperature increases because cohesive forces decrease with an increase of molecular thermal activity. The influence of the surrounding environment is due to the adhesive action of liquid molecules that they have at the interface.
What is molecular theory of surface tension?
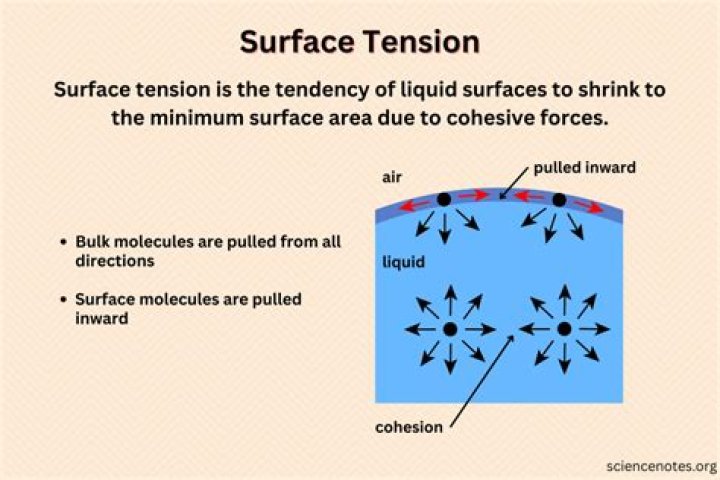
Surface tension has been well- explained by the molecular theory of matter. According to this theory, cohesive forces among liquid molecules are responsible for the phenomenon of surface tension. The molecules well inside the liquid are attracted equally in all directions by the other molecules.
What are the factors affecting angle of contact?
Two factors affecting the angle of contact: The nature of the liquid and the solid in contact. Impurities present in the liquid change the angle of contact.
What is the effect of surface tension?
Surface tension is what allows objects with a higher density than water such as razor blades and insects (e.g. water striders) to float on a water surface without becoming even partly submerged. … The net effect is the liquid behaves as if its surface were covered with a stretched elastic membrane.