What elements are in the same family
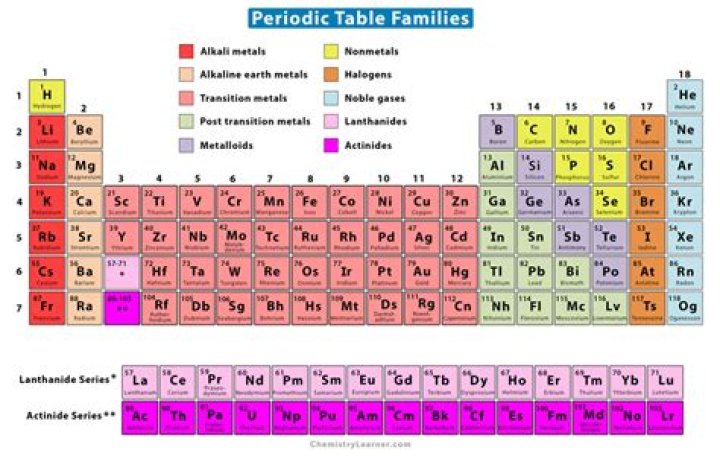
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties.
What elements are in the same family group?
The elements in the same group has similar number of valence electrons. They have identical number of electrons in their outermost shell. e.g. All the alkali metals in Group 1 have 1 valence electron, so they all tend to react the same way with other substances.
What are families of elements?
Related elements, including the noble gases, halogens, alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides. In addition, metals, nonmetals, and metalloids form loosely defined families. Other family designations—such as carbon family—are sometimes used.
What are 3 elements in the same family?
Lithium, Hydrogen, Sodium, or Oxygen, Sulfur, Selenium They are all in the same column.How are elements in the same family different?
Element families are elements that have the same number of valence electrons. Most element families are a single column of the periodic table, although the transition elements consist of several columns, plus the elements located below the main body of the table.
What 3 elements are in the same period?
HydrogenHeliumLithiumBerylliumNeonSodiumMagnesiumArgonPotassiumCalciumKryptonRubidiumStrontiumXenon
Which two elements are in the same period?
Elements that are in the same period have chemical properties that are not all that similar. Consider the first two members of period 3: sodium (Na) and magnesium (Mg). In reactions, they both tend to lose electrons (after all, they are metals), but sodium loses one electron, while magnesium loses two.
Why do elements in the same family have similar properties?
Because the members of each group in the main group elements has the same number of valence electrons, there are similar properties and similar trends in chemical reactions found in the group.What element is in period 4 Group 12?
zinc group element, any of the four chemical elements that constitute Group 12 (IIb) of the periodic table—namely, zinc (Zn), cadmium (Cd), mercury (Hg), and copernicium (Cn).
How many families of elements are there?5 Element Families The five element families combine similar element groups. So, even though there are numerous columns of transition metals on the periodic table, they all belong to the same family. The transition metals also include the lanthanides and actinides, which appear the main body of the table.
Article first time published onWhich set of elements has similar properties?
Elements in the same group of the periodic table have similar chemical properties. Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals.
What is similar about elements in the same group?
All the elements in one group have the same number of valence electrons. … These valence electrons are those involved in bonding with other atoms to form compounds. Since elements in a group have the same number of valence electrons, they behave similarly in chemistry.
What is similar about elements in the same period?
Elements in the same period have the same number of electron shells; moving across a period (so progressing from group to group), elements gain electrons and protons and become less metallic. This arrangement reflects the periodic recurrence of similar properties as the atomic number increases.
Why do elements in the same family generally have similar properties choose one as an example to support your reasoning?
Elements in the same family have similar properties because they have the same number of valence electrons. … So the nucleus has less of a effect on it’s electrons thus increasing the size of the atomic radius.
What element is in period 3 Group 13?
boron group element, any of the six chemical elements constituting Group 13 (IIIa) of the periodic table. The elements are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh).
What element is in Period 6 group 18?
Group118Atomic # Name55 Cs86 Rn
Which element is in group 14 Period 5?
Tin. Tin is a chemical element with the symbol Sn (for Latin: stannum) and atomic number 50. It is a main-group metal in group 14 of the periodic table.
What element is in group 16 Period 6?
oxygen group element, also called chalcogen, any of the six chemical elements making up Group 16 (VIa) of the periodic classification—namely, oxygen (O), sulfur (S), selenium (Se), tellurium (Te), polonium (Po), and livermorium (Lv).
Why don t elements in the same families share all of the same properties?
2) Why don’t elements in the same families share all of the same properties? There are other variables involved with these elements. For example, they have different sizes, different numbers of electrons, and different nuclear charges.
What characteristics do elements in the same group have in common select all that that apply?
What do elements in the same group have in common? They have similar chemical properties and the same number of valence electrons.
What family does neon belong to?
Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). The name comes from the fact that these elements are virtually unreactive towards other elements or compounds.
Which family is the element K in?
potassium (K), chemical element of Group 1 (Ia) of the periodic table, the alkali metal group, indispensable for both plant and animal life.
What are 3 elements that have similar properties?
In general, elements in the same column of the periodic table tend to have similar chemical properties. For instance, sodium, potassium, lithium, rubidium, and caesium all react exothermally with water, while copper, silver, and gold are all good conductors of electricity.
What two elements have similar characteristics?
Two elements have similiar characteristics when they belong to the same family that means that have placed in the same column of the periodic table. On the periodic table you can also find some chemical-phisic characteristics like electronegativity, density, melting point…
Which two elements are most likely to have similar physical properties?
Sodium and potassium share similar physical and chemical properties because they are in the same family or group called Alkali metals. Both will have the same valence electrons and they are both very reactive.
What do two elements in the same group share?
The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons. They are the electrons involved in chemical bonds with other elements. … Every element in the second column (group two) has two electrons in the outer shell.
What are the similar properties found among the members of same group?
The elements in a given group possesses similar electronic configuration, as the number of electrons in the respective outermost shells is the same. So, they have similar physical and chemical properties.