What is a trimeric G protein
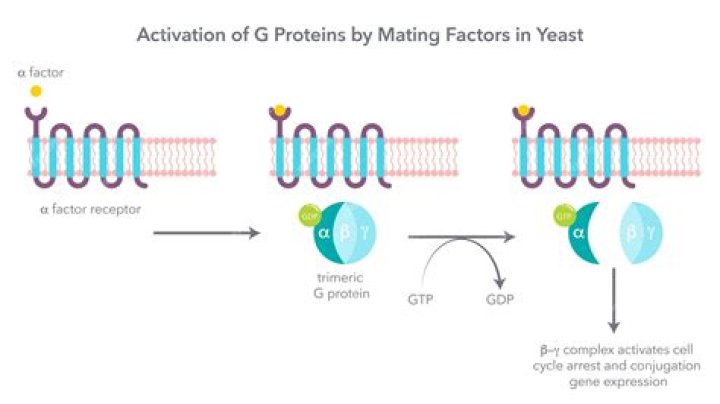
G proteins are attached to the cytosolic face of the plasma membrane, where they serve as relay proteins between the receptors and their target signalling proteins. Trimeric G proteins interact with 7TM receptors and are all heterotrimeric, having structurally different α, β and γ subunits.
What does an activated G-protein do?
Heterotrimeric G proteins couple the activation of heptahelical receptors at the cell surface to the intracellular signaling cascades that mediate the physiological responses to extracellular stimuli.
Is Transducin a typical trimeric G-protein?
Transducin (Td) is a prototypical hetero-trimeric G protein consisting of α-, β-, and γ-subunits. In the inactive state, the αβγ-trimer has GDP in the nucleotide-binding site of the α-subunit.
What are monomeric G proteins?
A monomeric G-protein (also known as small G protein or small GTPases, is the umbrella term for a family of signal transducing proteins. These comprise of: Rab, Arf, Ras, Ran and Rho, which are also vital in the growth of cells, as well as cell transport, motility, cytokinesis and cell differentiation.When a trimeric G-protein is activated it dissociates into two protein complexes What are these?
When stimulated by an activated receptor, the α subunit releases its bound GDP, allowing GTP to bind in its place. This exchange causes the trimer to dissociate into two activated components—an α subunit and a βγ complex (Figure 15-28).
Which G proteins do the different GPCRs use?
G proteins are divided into four subtypes, based on their Gα subunit: Gαs, Gαi/o, Gαq/11, and Gα12/13. Gβ and Gγ are closely associated and function as one unit: Gβγ. In general, GPCRs show a propensity to associate with certain subtypes, although most are able to signal through more than one subtype.
What do GTPases do?
3.2. 1 Introduction. Small GTPases are enzymes that catalyze the hydrolysis of guanosine triphosphate (GTP) to guanosine diphosphate (GDP). As the most well-known members, Ras GTPases play essential roles in regulating cell growth, cell differentiation, cell migration, and lipid vesicle trafficking.
Are G-proteins trimeric or monomeric GTPases?
G proteins are of two types- Monomeric and Trimeric G proteins respectively. Extracellular signalling molecules that bind to Enzyme-linked receptors are converted by monomeric G-proteins. Extracellular signalling molecules that bind to G-protein linked receptors are converted by trimeric G-proteins.What is the difference between monomeric and heterotrimeric G-proteins?
The biggest non-structural difference between heterotrimeric and monomeric G protein is that heterotrimeric proteins bind to their cell-surface receptors, called G protein-coupled receptors, directly. These G proteins are made up of alpha (α), beta (β) and gamma (γ) subunits.
What is the difference between heterotrimeric G-proteins and small G proteins?G protein can refer to two distinct families of proteins. Heterotrimeric G proteins, sometimes referred to as the “large” G proteins, are activated by G protein-coupled receptors and are made up of alpha (α), beta (β), and gamma (γ) subunits. “Small” G proteins (20-25kDa) belong to the Ras superfamily of small GTPases.
Article first time published onWhat happens when a trimeric G protein is activated?
Trimeric G Proteins are usually found coupled to a receptor protein and attached to the cytoplasmic face of the plasma membrane. In its inactive state the alpha sub-unit is bound to GDP, when the coupled receptor is activated this alpha sub-unit releases this GDP molecule. …
Is GPCR a GEF?
The GPCR, in essence, is a guanine-nucleotide exchange factor (GEF) for the Gα subunit. … GPCRs family is predicted to be present throughout the majority of sequenced eukaryotic genomes. Classically GPCRs activate a chemosensory transduction pathway through a change in the associated heterotrimeric G-protein activity.
What type of protein is rhodopsin?
Structurally, rhodopsin is classified as a chromoprotein (chromo is a Greek-derived root meaning “colour”). It is made up of opsin (a colourless protein) and 11-cis-retinal (11-cis-retinaldehyde), a pigmented molecule derived from vitamin A.
What attaches trimeric G proteins to the plasma membrane?
The GPCR can now activate a trimeric GTP-binding protein, or G protein (Figure 1). The G protein is composed of three protein subunits, Gα, Gβ, and Gγ. Both the Gα and Gγ subunits are post-translationally modified to have covalently attached lipid tails, which anchor the G protein to the plasma membrane.
Which is the subunit in the trimeric G-protein that interacts with GTP quizlet?
False: GRK, arrestin and clathrin is how the G-protein coupled RECEPTOR is inactivated. The trimeric G-protein is inactivated by GTP hydrolysis in the alpha subunit, which then binds to and inactivates the beta-gamma subunit.
When activated a G-protein has a structure that quizlet?
When activated, a G-protein has a structure thatChoose one:forms a transmembrane domain.is bound to GTP.is able to bind to extracellular signals. forms a complete complex with three subunits. What do the phosphorylated tyrosines on activated RTKs do?
How are GTPases deactivated?
Hydrolysis of GTP bound to an (active) G domain-GTPase leads to deactivation of the signaling/timer function of the enzyme. … GTPase activity serves as the shutoff mechanism for the signaling roles of GTPases by returning the active, GTP-bound protein to the inactive, GDP-bound state.
Where are GTPases found?
The different Rab GTPases are localized to the cytosolic face of specific intracellular membranes, where they function as regulators of distinct steps in membrane traffic pathways. In the GTP-bound form, the Rab GTPases recruit specific sets of effector proteins onto membranes.
How are GTPases regulated?
In order to speed up this alternation, the small GTPases are themselves regulated by guanine nucleotide exchange factors (GEFs), which assist in GDP dissociation, and also by GTPase activating proteins (GAPs) that stimulate GTP hydrolysis. …
How does GPCR activate G protein?
As their name implies, GPCRs interact with G proteins in the plasma membrane. When an external signaling molecule binds to a GPCR, it causes a conformational change in the GPCR. This change then triggers the interaction between the GPCR and a nearby G protein.
What does the G in GPCR stand for?
G protein-coupled receptor (GPCR), also called seven-transmembrane receptor or heptahelical receptor, protein located in the cell membrane that binds extracellular substances and transmits signals from these substances to an intracellular molecule called a G protein (guanine nucleotide-binding protein).
How many different G proteins are there?
G proteins are classified into four families according to their α subunit: Gi, Gs, G12/13, and Gq (Figure 1). The Gs and Gi families regulate adenylyl cyclase activity, while Gq activates phospholipase Cβ and G12/13 can activate small GTPase families (10).
How are monomeric G proteins activated?
proteins. These small proteins also operate as switches, which are “ON” when they bind GTP, and “OFF” after the GTP has been hydrolyzed to GDP (which in turn remains bound). Monomeric G-proteins are activated by proteins which induce a conformational change resulting in reduced affinity to GDP, and thus in GDP release.
How many heterotrimeric G proteins are there?
Heterotrimeric G protein structure Despite the size and diversity of the GPCR superfamily, these proteins interact with a relatively small number of G proteins to initiate intracellular signalling cascades. In humans, there are 21 Gα subunits encoded by 16 genes, 6 Gβ subunits encoded by 5 genes, and 12 Gγ subunits18.
What is monomeric receptor?
These data suggest that a monomeric receptor in a lipid bilayer is the minimal functional unit necessary for signaling, and that the cooperativity of agonist binding is due to G protein association with a receptor monomer and not receptor oligomerization.
How an activated trimeric G protein activates an effector protein?
(b) The change in conformation of the 7TM receptor on ligand binding brings about the binding of the trimeric G protein. (c) GTP binds and activates the α subunit, which becomes dissociated from the βγ complex. (d) The α subunit binds and activates target proteins, which also act as effectors and propagate the signal.
Is Ras trimeric?
Ras is structurally similar to the α subunit of trimeric G proteins. In fact, the GTP-binding domain of Gα is known as the Ras domain (Figure 35 ). Figure 35 Ras is structurally related to the α subunit of trimeric G proteins.
What type of membrane protein is typically associated with monomeric G proteins?
Ras proteins are monomeric guanine-nucleotide binding proteins, often termed small GTPases, that play essential roles in cell differentiation and proliferation. The term Ras superfamily is often used to describe a large family of over 100 members.
What types of heterotrimeric G proteins do you know?
Heterotrimeric G proteins consist of three distinct subunits, α, β and γ. These proteins couple the activation of diverse types of plasma membrane receptor to a variety of intracellular processes.
Why G protein is so named?
G-proteins are named for their ability to bind and hydrolyze the guanine nucleotide GTP.
What happens when GDP binds to the G protein?
Binding of GDP allows the α subunit to bind to the β and γ subunits to form an inactive trimer. Binding of an extracellular signal to a G-protein-coupled receptor allows the G-protein to bind to the receptor and causes GDP to be replaced with GTP (Figure 8.5A).