What is bromine water test
The bromine water test is a qualitative test, used to identify the alkene or alkane functional groups present in the compound. Alkene groups react with bromine water in the dark condition and undergo an addition reaction, to give a decolourized solution.
What color is a positive bromine test?
A positive test is indicated by: It turns red/brown if only saturated compounds are present. The degree of unsaturation of a compound can be determined by slowly adding bromine solution until the first red/brown color is observed. The more bromine solution required, the greater the unsaturation.
How does bromine water test for alkenes?
A simple test with bromine water can be used to tell the difference between an alkane and an alkene. An alkene will turn brown bromine water colourless as the bromine reacts with the carbon-carbon double bond. … The bromine water turns colourless confirming the presence of an alkene.
What color is a negative bromine test?
A negative test result is retention of the original color of the reagent, in this case the orange color (Figure 6.37b).What is a positive permanganate test?
The permanganate ion (MnO−4) is a deep purple color, and upon reduction converts to a brown precipitate (MnO2). Permanganate cannot react with aromatics, so is a good test to discern between alkenes and aromatics.
Is bromine soluble in water?
Bromine is a naturally occurring element that is a liquid at room temperature. It has a brownish-red color with a bleach-like odor, and it dissolves in water.
What are you testing for when you add bromine water to a lipid?
You will also test several lipids with bromine to determine the degree of unsaturation of the lipid. You may remember that alkenes react with bromine to give dibrominated compounds. The bromine adds to the double bond, and one bromine atom is placed on each side of the double bond.
What is the difference between bromine and bromine water?
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. … If an aqueous solution of bromine is used (“bromine water”), you get a mixture of products.How does bromine water test for alkanes?
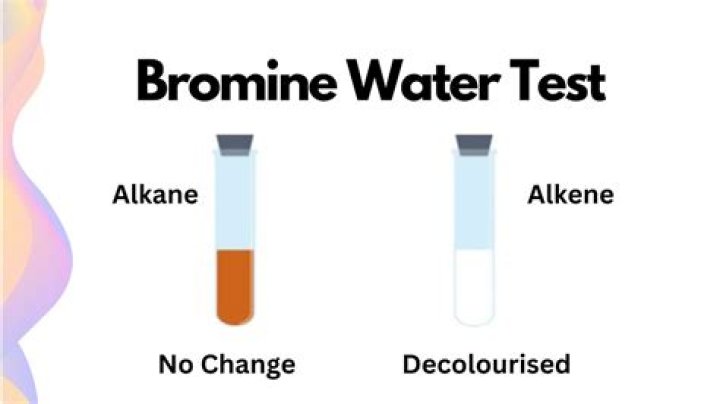
You can use bromine water, which is an orange solution, to distinguish between alkanes and alkenes. There is no change when bromine water is mixed with an alkane, but it turns colourless when mixed with an alkene.
What does baeyer's test indicate?-Baeyer’s test is basically a test for unsaturation. … Baeyer’s reagent is a strong oxidizing reagent which is used to identify the presence of double or triple bonds in a hydrocarbon. Thus it indicates the unsaturation of hydrocarbon compounds.
Article first time published onDoes ketone give bromine water test?
Aldehyde forms hydrate of aldehyde with water in presence of Bromine. Then bromine abstracts the hydride from hydrate of aldehyde, this step is not possible with ketones, because ketones will not have this hydride. This test plays a very important role in Carbohydrates chemistry to distinguish aldoses from ketoses.
How does potassium permanganate test work?
If the potassium manganate(VII) solution is acidified with dilute sulfuric acid, the purple solution becomes colorless. If the potassium manganate(VII) solution is made slightly alkaline (often by adding sodium carbonate solution), the purple solution first becomes dark green and then produces a dark brown precipitate.
What is the function of potassium permanganate test?
Potassium permanganate can be used to quantitatively determine the total oxidizable organic material in an aqueous sample. The value determined is known as the permanganate value.
What is the main purpose of huble's test?
(e) Huble’s Test: This test is used to know the degree of unsaturation in the given sample. Oils on reaction with Huble’s reagent fads the violet colour of iodine then it is unsaturated and if the colour persists then the given fat or oil is saturated.
Does benzene give bromine water test?
Benzene does not react with any electrophilic addition reactions hence it does undergo bromine water test, as they have delocalised pi bonds. Therefore, it does not decolourise the bromine water.
Which acid is used in salkowski test *?
Salkowski test is used to detect cholesterol in a solution. It is an important test used to detect cholesterol depending on the colors (distinct and clear colors) that yield from the reaction of cholesterol with concentrated sulfuric acid.
How do you make bromine water in a lab?
Dissolve the sodium bromine in the hydrochloric acid, mixing the compounds in a flask or beaker. Pour the mixture into the glass bottle. Add the bleach to the mixture in the bottle. Cap the bottle and swirl it gently to mix the ingredients.
Is bromine water safe?
Harmful if inhaled. Causes respiratory tract irritation and possible burns. Causes eye and skin irritation and possible burns. May cause digestive tract irritation with nausea, vomiting, and diarrhea.
Which of the following will not give bromine water test?
C3H8 (propane) is an alkane (saturated hydrocarbon) and hence it will not decolorize bromine water.
What is the use of bromine water?
In addition, bromine water is commonly used to test for the presence of an alkene which contains a double covalent bond, reacting with the bromine water, changing its color from an intense yellow to a colorless solution. Bromine water is also commonly used to check for the presence of an aldehyde group in compounds.
How does bromine react with water?
Reactions: Bromine. Bromine reacts with water to produce hypobromite, OBr-. The pH of the solution determines the position of the equilibrium. Bromine is not reactive towards oxygen or nitrogen but it will react ozone at -78°C to form the unstable compound bromine(IV) oxide.
What is bromine soluble?
Bromine is slightly soluble in water and highly soluble in many organic solvents, including carbon disulfide, carbon tetrachloride, acetic acid, and aliphatic alcohols. Bromine is not thought to persist in the environment.
Do alkanes react with bromine water?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot. … This has the effect of ‘saturating’ the molecule, and will turn an alkene into an alkane.
What happens when alkane reacts with bromine water?
there is no change when bromine water is mixed with an alkane. the bromine water becomes colourless when it is mixed with an alkene.
Is bromine water aqueous or liquid?
Bromine: It is soluble in water. Its aqueous solution is called bromine water.
How do you make bromine water?
This is chlorine water. Gradually, add potassium bromide (or sodium bromide) crystals to the prepared chlorine water and stir, until a deep orange coloured solution is formed. This is bromine water. solution to produce bromine water.
Does alcohol react with bromine water?
alcohol (by weight) and bromine in a molar ratio of two to one react at 25°, ethyl acetate and hy- drobromic acid are the sole reaction products. respect to free bromine. The tribromide ion formed during the reaction does not react with alcohol. … decrease of bromine is twice as fast as in the absence of aldehyde.
What is bare reagent?
Baeyer’s reagent is an alkaline solution of cold potassium permanganate, which is a powerful oxidant making this a redox reaction. Hence, option C is correct.
Do aldehydes give bromine water test?
The answer is yes. Aldehydes will decolourise bromine water.
How do you identify bromine?
A sensitive test for bromine is the reaction with fluorescein to give a deep red colour caused by bromination of the organic molecule, or by its reaction with fuchsine dyes in the presence of sulfurous acid, to give a deep blue colour.
How does bromine detect unsaturation?
Bromine solution is brown. In this test when bromine solution is added to the unsaturated hydrocarbon the brown colour disappears if the hydrocarbon is unsaturated. Bromine forms an addition product with the unsaturated hydrocarbon. Note: Decolourization of orange-red colour of bromine solution indicates unsaturation.

