What is electrochemical reaction in corrosion
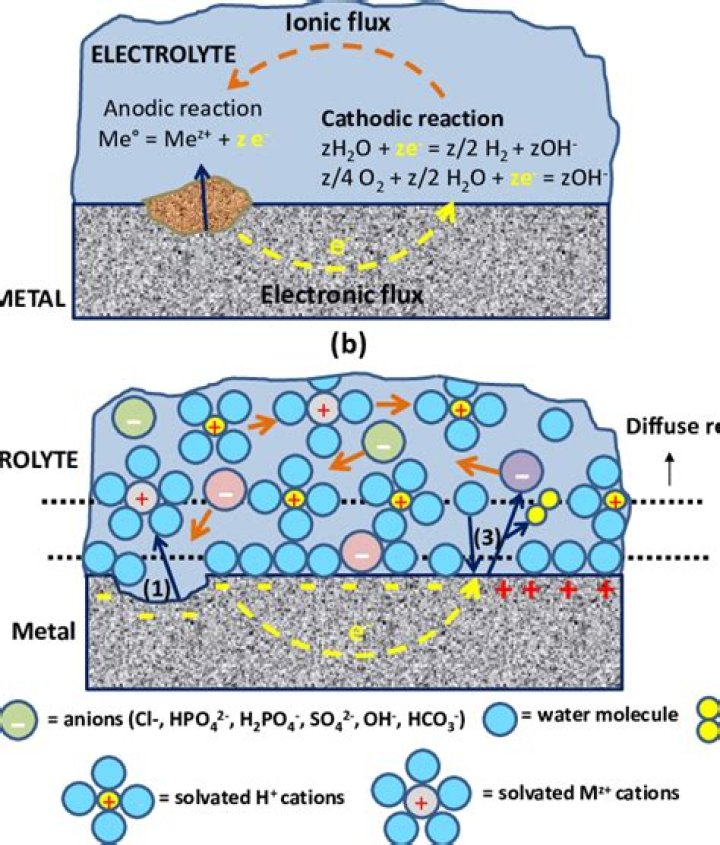
Electrochemical corrosion occurs when two dissimilar metals are present in an electrolytic medium. Sea water is an efficient electrolyte. Different parts of the same metal made dissimilar, by treatment, or a metal and its oxide are sufficiently dissimilar to create such corrosion as shown in Fig. 14.16.
Why corrosion is an electrochemical reaction?
Corrosion is an electrochemical method by which materials are deteriorated. In many cases—and especially when liquids are present—it involves chemistry. During corrosion, electrons from distinct areas of a metal surface flow to alternative areas through an atmosphere capable of conducting ions.
What is electrochemical reaction explain it with example?
It is a reaction which occurs with absorption of electrical energy. Example: Acidulated water breaks into hydrogen and oxygen. 2 H X 2 O → Electricity 2 H X 2 + O X 2.
How is corrosion an electrochemical process?
The corrosion rate is enhanced by an electrochemical process in which a water droplet becomes a voltaic cell in contact with the metal, oxidizing the iron. Within the droplet, the hydroxide ions can move inward to react with the iron(II) ions moving from the oxidation region. Iron(II) hydroxide is precipitated.What are electrochemical methods of corrosion protection?
Corrosion can be prevented by application of electrochemistry principles. This basically falls into two distinct areas, sacrificial anodes and cathodic protection by impressed currents. Sacrificial Anodes.
What is electrochemical or wet corrosion?
Wet corrosion refers to the degradation and/or rust formation on a metal surface that occurs due to the generation of a reactive electrochemical cell. Wet corrosion can damage metal structures and equipment, including stainless steel.
What is electrochemical corrosion and galvanic corrosion?
Galvanic corrosion, also known as bimetallic corrosion, is an electrochemical process whereby one metal corrodes in preference to another metal that it is in contact with through an electrolyte. Galvanic corrosion occurs when two dissimilar metals are immersed in a conductive solution and are electrically connected.
Is corrosion an electrochemical phenomenon?
The chemistry of corrosion of iron is essentially an electrochemical phenomenon. … It can, therefore, be referred to as an electrochemical reaction. In the process of corrosion, due to the presence of air and moisture, oxidation takes place at a particular spot of an object made of iron. That spot behaves as the anode.What are the types of electrochemical corrosion?
- Microbiologically Induced Corrosion (MIC) …
- Crevice Corrosion. …
- Galvanic Corrosion. …
- Uniform Corrosion. …
- Pitting Corrosion. …
- Electrochemical Noise (ECN) …
- Electrochemical Impedance Spectroscopy (EIS) …
- Linear Sweep Voltammetry (LSV)
Rusting, the formation of iron oxides, is a well-known example of electrochemical corrosion. This type of damage typically produces oxide(s) or salt(s) of the original metal and results in a distinctive orange colouration.
Article first time published onWhat is electrochemical reaction Class 10?
Electrochemical reaction: The chemical reactions which proceed with the absorption of electric energy are called electrochemical reactions. Example: Electric current.
What is electrochemical reaction Class 8?
Ans: – Chemical reaction in which electrical energy is consumed or produced are known as electrochemical reaction.
What is meant by electrochemical method?
Electrochemical methods remove and recover heavy metals based on the principle that metals become precipitated in their elemental form on the solid electrodes when the potential or electricity is applied on the electrode.
What are the advantages of electrochemical method?
Electrochemical techniques have their advantages because of their simplicity, low cost and speed. The only condition for this method of monitoring enzyme kinetics is that direct electron transfer must be possible or able to be achieved by mediators [14–18].
What are the examples of electrochemical methods of analysis?
Commonly used electrochemical techniques include amperometry, potentiometry, conductometry, voltammetry, and galvanic cell.
What is electrochemical corrosion monitoring?
Electrochemical corrosion potential is an effective method for corrosion monitoring and detection of components in a nuclear power plant. On one hand, the electrochemical corrosion potential could provide a guide to determine the amount of an oxidation/reduction reaction which occurs in metal.
How is electrochemical corrosion measured?
- Linear polarization resistance (LPR) measurements.
- Potentiodynamic polarization curves.
- Electrochemical potentiokinetic reactivation (EPR) measurements for intergranular corrosion.
- Current vs time curves (at a given potential)
What is electrochemical measurement?
Electrochemical Measurement Method In electrochemical experiments, an electrochemical reaction is caused in a sample in a solution in a cell, and the electrical response is measured. … In general electrochemical measurements, three electrodes are immersed in a test solution (electrolyte).
What is electrochemical nature of aqueous corrosion?
Aqueous Corrosion (corrosion in water media) is electrochemical in nature. This means that the corrosion reaction involves electrons (e-). … Aqueous Corrosion involves the loss of metal to the aqueous phase. When this occurs, the metal releases a number of electrons (e-) into the Metal/Alloy.
What is electrochemical corrosion explain the mechanism of electrochemical corrosion by evolution of hydrogen gas?
Evolution of Hydrogen Electrochemical corrosion with evolution of H2 occurs in acidic Environment At anode Fe →Fe2+ + 2e-(oxidation /corrosion) At cathode 2H+ + + 2e-→H2 Overall reaction Fe +2H+ →Fe Explanation (1) Iron tank which acts as an anode undergoes corrosion as Fe atoms from the tank pass into the acidic …
What are the two partial reactions electrochemical corrosion reaction consists of?
Corrosion reactions of metallic materials are of electrochemical nature, that is, the reaction can be divided into an oxidation reaction (anodic reaction) and a reduction reaction (cathodic reaction).
What is electrochemical series?
Electrochemical series is a series of chemical elements arranged in order of their standard electrode potentials.
How is rusting of iron an electrochemical phenomenon?
Answer: Rusting of iron is a electrochemical phenomenon because rusting involves the formation of electrochemical cells on the surface of iron in which water act as the electrolyte. … Thus iron loses electrons and get converted to rust and lose its shine.
What is corrosion give mechanism of rusting of iron?
Rusting of iron refers to the formation of rust, a mixture of iron oxides, on the surface of iron objects or structures. This rust is formed from a redox reaction between oxygen and iron in an environment containing water (such as air containing high levels of moisture).
What protects iron corrosion?
Galvanize: Galvanizing coats iron or steel in zinc to protect from rust. Zinc corrodes at a much slower rate than iron or steel, so it’s highly effective for slowing rust. Blueing: This process creates a layer of magnetite over the metal to prevent rust.
Which of the following is true about electrochemical corrosion?
Which of the following true about Electrochemical corrosion? A. Electrochemical corrosion of metals occurs when proton from atoms at the surface of the metal are transferred to a suitable proton acceptor or depolarizer.
What is electrochemical reaction Class 12?
Chemical reactions that involve the input or generation of electric currents are called electrochemical reactions. … Production of chemical change by electrical energy i.e. the phenomenon of electrolysis. Conversion of chemical energy into electrical energy.
What are photochemical and electrochemical reactions give an example in each case?
(a) Photochemical reaction: It is a reaction which occurs with the absorption of light energy. (b) Electrochemical reaction: It is a reaction which occurs with the absorption of electrical energy. Example: Acidulated water breaks into hydrogen and oxygen.
What is the difference between chemical reaction and electro chemical reaction?
Answer and Explanation: Chemical reactions can cause chemical changes while electrochemical reactions are used to generate electric charges and chemical changes occur.
What is electrochemical reaction Class 9?
b) A reaction that occurs with the absorption of electrical energy is called an electrochemical reaction. Ex: Acidulated water breaks into hydrogen and oxygen.
What is meant by thermochemical reactions?
Thermochemistry is the study of the energy and heat associated with chemical reactions and/or physical transformations. A reaction may release or absorb energy, and a phase change may do the same, such as in melting and boiling.