What is special about the structure of water
Water molecules are polar, so they form hydrogen bonds. This gives water unique properties, such as a relatively high boiling point, high specific heat, cohesion, adhesion and density.
Why is water's structure special?
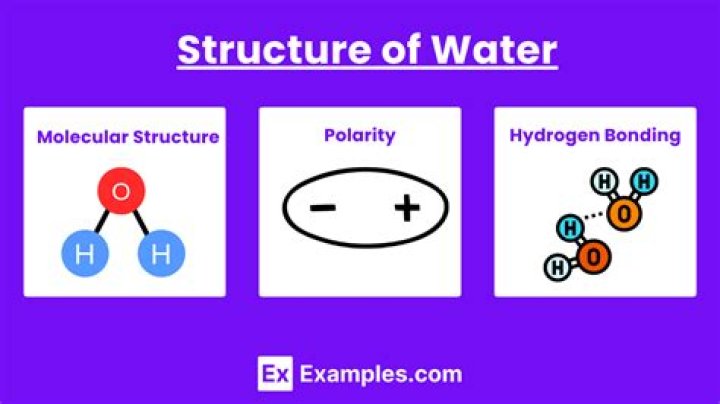
The key to understanding water’s chemical behavior is its molecular structure. A water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons.
What are the special properties of water?
- Water is polar. …
- Water is an excellent solvent. …
- Water has high heat capacity. …
- Water has high heat of vaporization. …
- Water has cohesive and adhesive properties. …
- Water is less dense as a solid than as a liquid.
What is the structure of water?
A water molecule is made up of two hydrogen atoms and one oxygen atom. A single oxygen atom contains six electrons in its outer shell, which can hold a total of eight electrons. When two hydrogen atoms are bound to an oxygen atom, the outer electron shell of oxygen is filled.How does the structure of water relate to its function?
This molecular structure gives the water molecule polarity, or a lopsided electrical charge that attracts other atoms. The end of the molecule with the two hydrogen atoms is positively charged. … Water’s polarity allows it to dissolve other polar substances very easily.
Does water have a structure?
It possesses a regular crystalline structure based on the molecular structure of water, which consists of a single oxygen atom covalently bonded to two hydrogen atoms: H-O-H. The fact the density of ice is less than that of liquid water’s has the important consequence that ice floats.
Why does h20 have bent structure?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). The molecule adopts a bent structure because of the two lone pairs of electrons on the oxygen atom. …
How was the structure of water determined?
It was the chemist Henry Cavendish (1731 – 1810), who discovered the composition of water, when he experimented with hydrogen and oxygen and mixed these elements together to create an explosion (oxyhydrogen effect). In 1811 the Italian physician Amedeo Avogadro finally found the H2O formula for water.Why is water special in regards to its structure and intermolecular forces?
Water has some unusual properties due to its hydrogen bonds. One property is cohesion, the tendency for water molecules to stick together. The cohesive forces between water molecules are responsible for the phenomenon known as surface tension.
What about the structure of water molecules determines why water on Earth is most often found in this liquid form?Water forms a liquid instead of a gas because oxygen is more electronegative than the surrounding elements, with the exception of fluorine. Oxygen attracts electrons much more strongly than does hydrogen, resulting in a partial positive charge on the hydrogen atoms and a partial negative charge on the oxygen atom.
Article first time published onWhy is h20 not linear?
In water, the oxygen atom has two lone pairs. These two lone pairs repel the hydrogen-oxygen bonded pairs so much that the molecule is at its lowest energy arrangement when the H-O-H bond angle is 104.5 degrees. As a result, the water molecule can be classified as non-linear.
Can water change its structure?
Water is a very strong molecule. When inside the brain it is a solvent for electrolytes and neurotransmitters and during this time it does not change much from it’s H2O structure. The only thing that can change H2O is extremely high heat or near absolute zero cold, which the brain has no capability in.
Why is water unique from other liquids?
In the liquid form, the hydrogen atoms of one water molecule are attracted to the oxygen atom of another molecule. Each water molecule can form up to four of these hydrogen bonds and, collectively, they give water a cohesiveness unique in liquids.
How will you describe the molecular structure of water in solid form?
When water freezes, water molecules form a crystalline structure maintained by hydrogen bonding. Solid water, or ice, is less dense than liquid water. Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart, which lowers the density.
How does the structure of water affect its ability to act as a solvent?
Water molecules have a polar arrangement of oxygen and hydrogen atoms—one side (hydrogen) has a positive electrical charge and the other side (oxygen) had a negative charge. This allows the water molecule to become attracted to many other different types of molecules.
Why is water molecule at an angle?
The angle between hydrogen atoms in the water molecule is approximate ${ 105 }^{ ° }$ which is the lowest energy configuration for the system. In the water molecule, the resulting angle occurs due to the oxygen atom having two sets of lone pair electrons with interacting repulsive fields.
Why are water molecules V shaped?
It contains two pairs of bonded electrons and two unshared lone pairs. Due to itsshape, and polarity, water molecules are greatly attracted to one another. In water , 105 degree bond in the angles b/w the hydrogen and oxygen . Because of 2 atoms of hydrogen ana 1 atom of oxygen water molecule is V-shaped.
What is the shape of water chemistry?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.

