What is stilbene dibromide used for
Stilbene is used in manufacture of dyes and optical brighteners, and also as a phosphor and a scintillator. Stilbene is one of the gain mediums used in dye lasers.
What color is stilbene dibromide?
NamesMolar mass340.058 g·mol−1AppearanceWhite solidMelting point241 °C (466 °F; 514 K)Dipole moment0.4-0.9
What is bromination of stilbene?
Bromination of trans-stilbene and the formation of stilbene dibromide is a rapid and. stereospecific reaction that involves the nucleophilic attack of an alkene to a bromine and a. bromide ion attack on a cation intermediate formed through the nucleophilic attack.
Is meso stilbene dibromide toxic?
Causes eye irritation and possible burns. Skin: Causes skin irritation and possible burns. Ingestion: May cause irritation of the digestive tract and possible burns. Inhalation: May cause severe irritation of the respiratory tract with possible burns.Why does the reaction using e stilbene produce a meso product?
The cyclic bromonium ion takes on a positive charge and by anti-addition the second bromine (negatively charged) attacks from the back of the cyclic compound and bonds to either carbon on the opposite side of the other bromine. This creates a meso-stilbene which is 100% formed.
What is the molar mass of stilbene dibromide?
PubChem CID53432511Molecular FormulaC14H12Br2-2Molecular Weight340.05Parent CompoundCID 11502 (Stilbene)Component CompoundsCID 11502 (Stilbene) CID 260 (Hydrogen bromide)
What was the solvent for the bromination of stilbene?
Bromine and chlorine readily undergo addition reactions with alkenes. This practical involves the bromination of trans-stilbene using a 10% bromine dichloromethane solution.
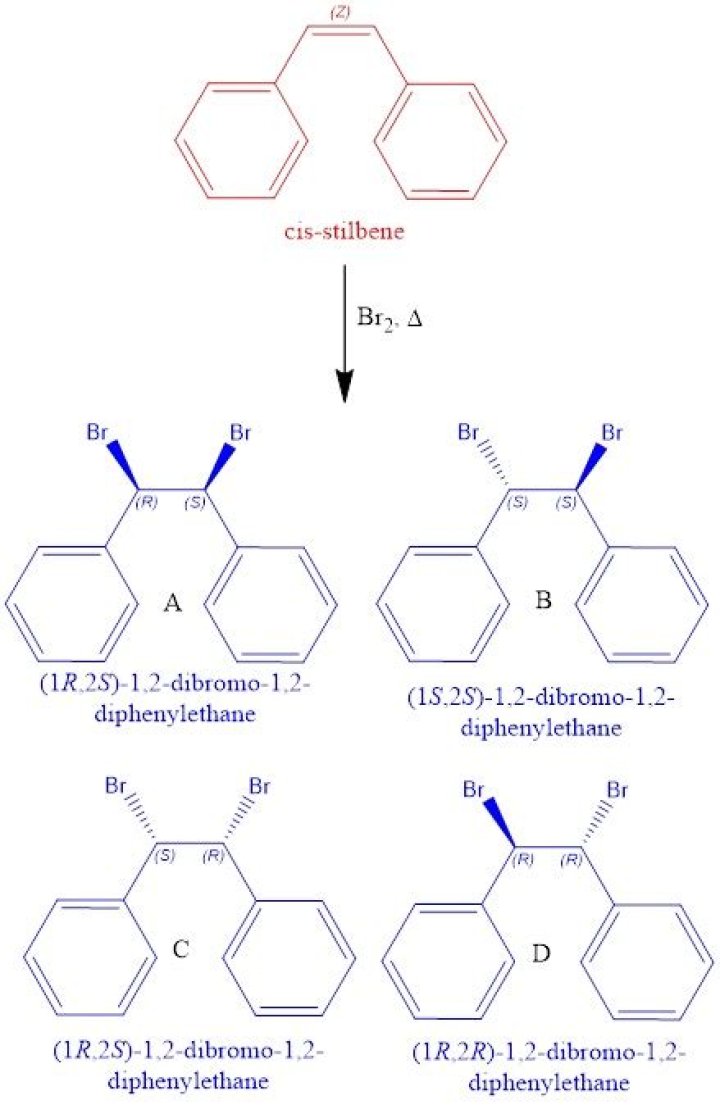
How many possible stereoisomers exist for stilbene dibromide?
There are three stereoisomers of stilbene dibromide namely (1R, 2S)-1,2-dibromo-1,2-diphenylethane, (1R, 2R)-1,2-dibromo-1,2-diphenylethane and (1S,…What is the boiling point of meso stilbene dibromide?
Density:1.6±0.1 g/cm3Boiling Point:323.8±37.0 °C at 760 mmHgVapour Pressure:0.0±0.7 mmHg at 25°CEnthalpy of Vaporization:54.3±3.0 kJ/molFlash Point:173.2±25.7 °C
Is meso stilbene optically active?Is Stilbene dibromide, Product 106550, optically active? Generally, chiral molecules are optically active, rotating polarized light. Meso compounds, such as 106550, are unique in that there are chiral carbons in the molecule yet they are not optically active.
Article first time published onIs stilbene an alkene?
Common name for trans-1,2-diphenylethylene, aka, stilbene, or more specifically, (E)-stilbene, is the alkene, ethene with two phenyl groups on either carbon of the parent chain. The name was derived from the Greek word stilbos, which means shining.
What are the products of bromination of e stilbene?
Bromination of trans-stilbene produces predominantly meso-1,2-dibromo-1,2-diphenylethane (sometimes called meso-stilbene dibromide), in line with a mechanism involving a cyclic bromonium ion intermediate of a typical electrophilic bromine addition reaction; cis-stilbene yields a racemic mixture of the two enantiomers …
What is the purpose of bromination?
Bromination of Alkenes Gives anti Products It’s a family of reactions which proceed through 1) attack of an alkene upon an acid, forming a free carbocation, and 2) attack of a nucleophile upon the carbocation.
Why is pyridinium Perbromide used in this experiment?
In these two bromination reactions, pyridinium tribromide will be used as the source of bromine. This solid reagent is safer and easier to handle than elemental bromine, which is a highly corrosive liquid.
What is the melting point of cis stilbene?
PubChem CID5356785DescriptionCis-stilbene is an oily yellow liquid. Freezing point 5°C. Insoluble in water. Soluble in ethanol. Unstable relative to the trans isomer, which is a solid with a melting point of 122-124°C. CAMEO Chemicals Cis-stilbene is the cis-isomer of stilbene. ChEBI
Why is the melting point of Z stilbene so much lower than e stilbene?
(Z)-Stilbene is sterically hindered and less stable because the steric interactions force the aromatic rings 43° out-of-plane and prevent conjugation. (Z)-Stilbene has a melting point of 5–6 °C (41–43 °F), while (E)-stilbene melts around 125 °C (257 °F), illustrating that the two compounds are quite different.
Is pyridinium tribromide carcinogenic?
Not available. Carcinogenicity: CAS# 39416-48-3: Not listed by ACGIH, IARC, NTP, or CA Prop 65.
What is the melting point of Diphenylacetylene?
Density0.9900g/mLColorBrown to YellowMelting Point58.0°C to 61.0°CBoiling Point170.0°C (19.0 mmHg)Assay Percent Range98.5% min. (GC)
What is the density of pyridinium tribromide?
Solubility0.17 g/l (20 °C)Flash Point118 °CRefractive Index1.457 (25 °C, 589 nm)Density1.03 g/cm3 (20 °C)Ignition Point215 °C
Do meso compounds have enantiomers?
Just to make things clear, a meso compound is a molecule that has chiral centers but also has an internal plane of symmetry. This renders the molecule achiral: it does not have an enantiomer, and it does not rotate plane polarized light .
What is the melting point of 1/2-dibromo-1,2-diphenylethane?
Density1.613 g/cm3Melting Point235-241ºCMolecular FormulaC14H12Br2Molecular Weight340.05300Flash Point173.2ºC
What is the melting point range of meso 1/2-dibromo-1,2-diphenylethane?
meso-1,2-Dibromo-1,2-diphenylethane is used to study the reaction of ± and meso-SBr2 with 9-substituted fluorenide ions in dimethyl sulfoxide. Melting Point : 241° C (dec.)
What kind of reaction is bromination of e stilbene?
The reaction of bromine with (E)-stilbene is a classic reaction performed as an example of an electrophilic addition reaction of alkenes. The reaction is stereospecific through an anti addition to the double bond. The traditional reaction uses a bromine solution in methylene chloride, which has very high toxcity.
What is meant by meso compound?
A meso compound or meso isomer is a non-optically active member of a set of stereoisomers, at least two of which are optically active. This means that despite containing two or more stereogenic centers, the molecule is not chiral.
What is a meso sugar?
Introduction. In general, a meso compound should contain two or more identical substituted stereocenters. Also, it has an internal symmetry plane that divides the compound in half. These two halves reflect each other by the internal mirror. … Cyclic compounds may also be meso.
Does bromination follow Markovnikov's rule?
When the two carbons of the halonium ion are not the same, more positive charge ends up on the carbon attached to the greatest number of carbons. Or, the bromine ends up on the carbon with the most hydrogens. That is, the addition of HOBr follows Markovnikov’s rule.
What products would you expect from bromination of styrene?
Bromination of styrene in the presence of a large excess of acetate or nitrate gives only two products in each case, the 0,8-dibromide and the -acetoxy-8-bromide or a-nitrato-b-bromide respectively.
Is pyridinium Perbromide a strong base?
it is a strong base it is a sterically hindered base and therefore well suited to perform an E2 elimination it is a solid form of bromine with less toxicity tnan bromine it is not reactive with a Ikene QUESTION 2 Tne addition of Brz to a alkene is syn accition process True False QUESTION 3 What is the role of potassium …
What are some advantages of using pyridinium bromide in place of molecular bromine?
- Apart from being a source of bromine in the reaction, its solid structure makes it handier. …
- Liquid bromine can cause spillage or damage to the skin because of its corrosive nature, whereas pyridinium tribromide is much safer to handle.
What safety precautions should be taken when working with pyridinium tribromide?
Avoid contact with skin and eyes. Control personal contact by using protective equipment. Use dry clean up procedures and avoid generating dust. Place in a suitable, labelled container for waste disposal.