What is the shape of the 2p atomic orbital
Each 2p orbital has two lobes. There is a planar node normal to the axis of the orbital (so the 2px orbital has a yz nodal plane, for instance). The higher p-orbitals ( 3p, 4p, 5p, 6p, and 7p) are more complex still since they have spherical nodes as well.
What is the shape of the 2p orbital dumbbell?
p-orbital is dumb-bell shaped.
How many atomic orbitals are in 2p?
However, there are three orbitals in the 2p subshell.
Is 2p an atomic orbital?
The shapes of the first five atomic orbitals are: 1s, 2s, 2px, 2py, and 2pz.What overall shape do 2p and 3p orbitals have?
The similarity of these two orbital is both orbitals have same shape two-lobbed shape.
Is P orbital sphere?
The p orbital appears as a dumbbell – a spherical shape like the s orbital cut in half. As the atomic nucleus spins, individual protons also spin. There are two times during a rotation that three protons align – 90° and 270° (below).
Which atomic orbital is dumbbell in shape?
The s orbital is a spherical shape. The p orbital is a dumbbell shape.
What is orbital shape?
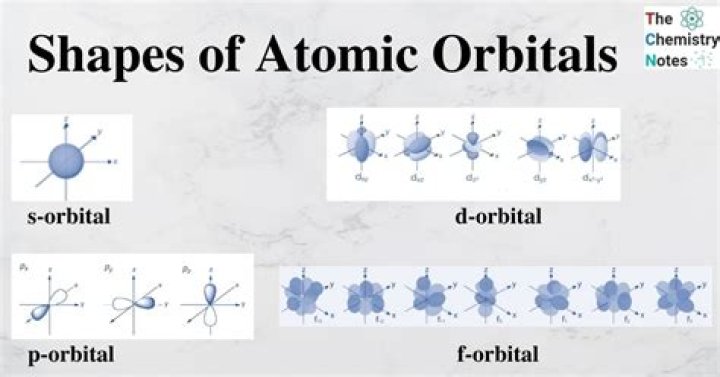
An s-orbital is spherical with the nucleus at its centre, a p-orbitals is dumbbell-shaped and four of the five d orbitals are cloverleaf shaped. The fifth d orbital is shaped like an elongated dumbbell with a doughnut around its middle. The orbitals in an atom are organized into different layers or electron shells.What is 2p chemistry?
This number indicates how many orbitals there are and thus how many electrons can reside in each atom. Orbitals that have the same or identical energy levels are referred to as degenerate. An example is the 2p orbital: 2px has the same energy level as 2py.
How is a 2p orbital different from a 2s orbital?The 2 s and 2 p orbitals differ in shape, number, and energy. A 2 s orbital is spherical, and there is only one of them. A 2 p orbital is dumbbell-shaped, and there are three of them oriented on the x, y, and z axes. The 2 p orbitals have higher energy than the 2 s orbital.
Article first time published onWhat overall shape do the p orbitals have?
Explanation: All p orbitals have a characteristic dumbbell shape with a nodal plane perpendicular to the orbital axis.
How many orbitals are in the 2p sub level?
There are three orbitals in the 2p sublevel. These three orbitals can hold two electrons each for a total of six electrons.
What is the shape of a 3p atomic orbital?
The shape of a 3p orbital resembles dumbbell-shape. There are four different types of orbitals, each with a different shape, denoted by the letters s, p, d, and f. An s-orbital is spherical with the nucleus at its centre, a p-orbitals is dumbbell-shaped and four of the five d orbitals are cloverleaf shaped.
What is the difference between 2p and 3p?
There is no difference in the shape of 2p and 3p orbitals as for both the azimuthal quantum number is same, which determines the shape of orbital.
What is the difference between 3p and 2p?
The 3p orbital has two nodal planes, while the 2p orbital has only one. The 3p orbital is farther from the nucleus than the 2p orbital The 3p orbital is a different shape than the 2p orbital.
What is dumbbell shape?
Dumbbell Shapes Pro dumbbells have a short, cylindrical handle and pancake ends, which are circular with a flat edge.
Which of the following is dumbbell shaped?
P orbital is dumb-bell shaped.
What is the shape of d and f orbitals?
Orbitals with ℓ=1 are p orbitals and contain a nodal plane that includes the nucleus, giving rise to a dumbbell shape. Orbitals with ℓ=2 are d orbitals and have more complex shapes with at least two nodal surfaces. Orbitals with ℓ=3 are f orbitals, which are still more complex.
How are p orbitals different?
The s orbital is spherical, while the p orbital is shaped like a dumbbell. Due to these shapes, the s orbital has only one orientation, while the p orbital has three degenerate orientations ( x , y , and z ), each of which can hold up to two electrons.
How many orbitals are in P?
The p sublevel has 3 orbitals, so can contain 6 electrons max.
What is difference between s and p orbital?
The s orbitals are atomic orbitals and the shape of s orbital is spherical. The p orbital is also an atomic orbital and the shape of p orbital is dumbbell shape. S orbitals have the lowest energy levels. The energy levels of p orbitals are higher as compared to that of p orbitals.
What are 1s 2s 2p?
1s will be filled first, with the maximum of 2 electrons. • 2s will be filled next, with the maximum of 2 electrons. • 2p will be filled next, with the maximum of 6 electrons.
How do you find atomic orbitals?
- The principal quantum number, n , determines the size of the orbital.
- The secondary quantum number, l , determines the shape.
- For each value of n , there is one orbital for which l=0 .
- The higher the value of n , the larger the sphere.
What are the 4 atomic orbitals?
Current theory suggests that electrons are housed in orbitals. An orbital is a region of space where there is a high probability of finding an electron. There are four basic types of orbitals: s, p, d, and f. An s orbital has a spherical shape and can hold two electrons.
What is the shape of orbital with L 2 and L 3?
The orbitals with l= 2 are the d orbitals which have complex shapes with at least two nodal surfaces. The orbitals with l= 3 are called the f orbitals that are more complex.
Which is bigger 2s or 2p orbital?
The radial distribution functions for a 2s and 2p orbital are illustrated below. On average, the 2s electrons will be at a slightly greater distance from the nucleus than the 2p electrons. However, the 2s electrons have a higher probability of being closer to the nucleus due to the inner peak.
Do 2s and 2p have the same shape?
The 2 s and 2 p orbitals differ in shape, number, and energy. A 2 s orbital is spherical, and there is only one of them. A 2 p orbital is dumbbell-shaped, and there are three of them oriented on the x, y, and z axes. … 2p has dumbbell shaped ( it has no radial node ).
Why is 2p higher than 2s?
2p has higher energy level because the negatively charged electron experiences less of an effective nuclear charge than the 2s electron.
How many total orbitals are within the 2s and 2p?
The second energy level will thus have a total of 4 orbitals. These orbitals will be distributed on two subshells, the s-subshell and the p-subshell.
How many orbitals are in the 2p sublevel quizlet?
The 2p sublevel contains three orbitals, with one arrow pointing upward in the first orbital, one arrow pointing upward in the second orbital, and one arrow pointing upward in the third orbital.
How many orbitals are in each level?
Recall that the four different sublevels each consist of a different number of orbitals. The s sublevel has one orbital, the p sublevel has three orbitals, the d sublevel has five orbitals, and the f sublevel has seven orbitals.

