What is the trend of reactivity
Period – reactivity decreases as you go from left to right across a period. The farther to the left and down the periodic chart you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity.
What is the trend in reactivity down the group?
Reactivity increases down the group. This is because the outer electron in each atom is further away from the nucleus as you go down the group and so the electrostatic forces between the nucleus and the outer electron are weaker. This means electrons are more easily given up in a reaction.
What is the trend of reactivity in group 1?
The reactivity of Group 1 elements increases as you go down the group because: the atoms get larger as you go down the group. the outer electron gets further from the nucleus as you go down the group.
What is the trend of reactivity of metal ions?
In the reactivity series, as we move from bottom to top, the reactivity of metals increases. Metals present at the top of the series can lose electrons more readily to form positive ions and corrode or tarnish more readily.What is the order of reactivity?
In order of decreasing reactivity, the metals potassium, sodium, lithium, calcium, magnesium, zinc, iron and copper can be put in order of their reactivity from their reactions with water and dilute acids.
Why does reactivity increase or decrease down a group?
As we go down the group, the atom gets bigger. The bigger the atom, the further away the last electron. Therefore, the attraction between the nucleus and the last electron gets weaker. This makes it easier for the atom to give up the electron which increases its reactivity.
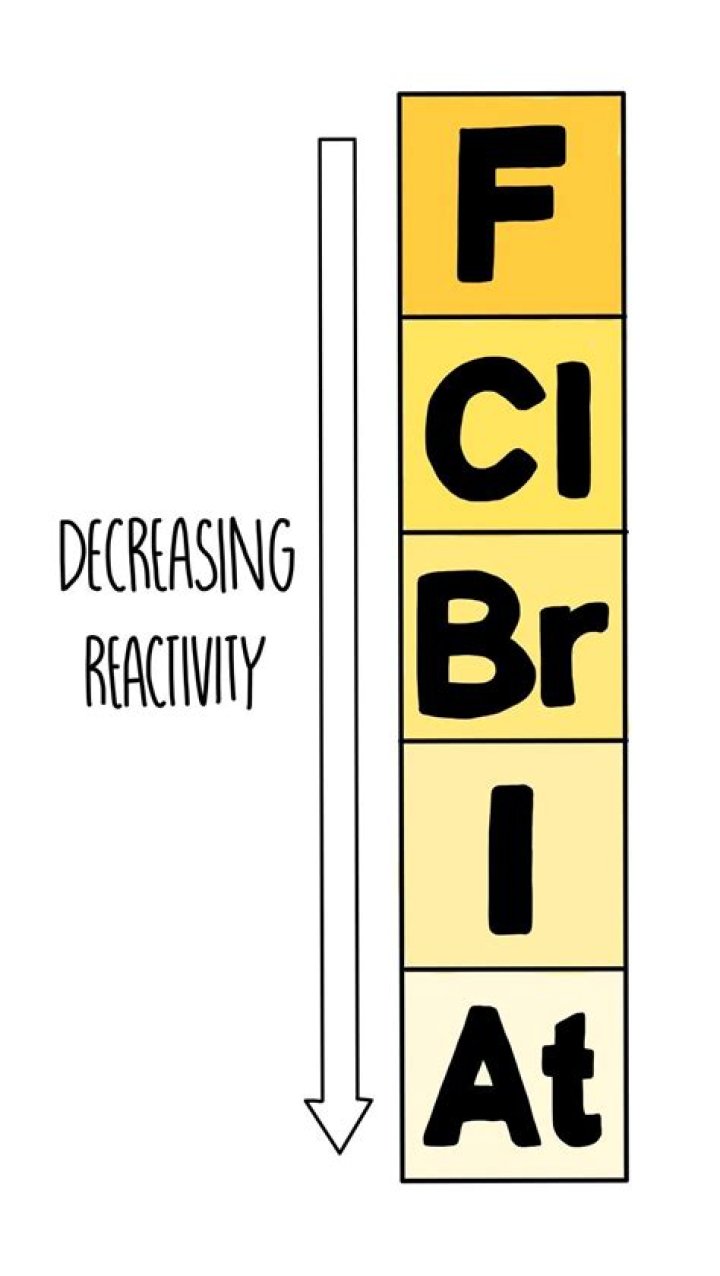
What is the trend of reactivity in Group 7?
Reactivity decreases down the group. This is because group 7 elements react by gaining an electron. As you move down the group, the amount of electron shielding increases, meaning that the electron is less attracted to the nucleus.
What is metal reactivity?
Reactive metal is a group of metal elements that can form a reaction with acids, water, mineral acids and powerful oxidizing acids. … Starting at the bottom of the series and going up, the reactivity of metals increases and loses electrons to create positive ions.Does reactivity increase down group 3?
Group – reactivity decreases as you go down the group.
Why is the trend in reactivity different for metals and nonmetals?Since the ionization energy decreases going down a group (or increases going up a group), the increased ability for metals lower in a group to lose electrons makes them more reactive. … Nonmetals tend to gain electrons in chemical reactions and have a high attraction for electrons within a compound.
Article first time published onWhat is the trend in reactivity in Group 2?
The reactivity of the group 2 elements increase as you go down the group. This is because, as explained previously, it is much easier to remove an outer shell electron as you go further down the group (lower ionisation energies).
How does reactivity change down Group 2?
As you progress down Group 2, the reactivity increases. This is due to a decrease in ionisation energy as you progress down the group. As it requires less energy to form the ions, the reactivity increases.
What is the reactivity of Group 2 elements?
Reactivity of Group II elements increases down the group. This can be explained by the increase in ease at losing two outer electrons as we descend the group. The loss of electrons becomes easier due to the decreasing ionisation energy required.
Which of the following is arranged according increasing reactivity?
According to the increasing order of there reactivity, the given elements are arranged as Copper<Zinc<Aluminium<Magnesium<Sodium.
How do you remember reactivity series?
Elements of Reactivity SeriesSentence to Remember the SeriesZn (Zinc)ZebraFe (Iron)InPb (Lead)Lead &H (Hydrogen)Hydrogen
What is the list of reactivity called?
The reactivity series is a list of metals ranked in order of decreasing reactivity, which is usually determined by the ability to displace hydrogen gas from water and acid solutions.
What are the trends in group 1 and 7?
The non-metal elements in Group 7 – known as the halogens – get less reactive as you go down the group. This is the opposite trend to that seen in the alkali metals in Group 1 of the periodic table . Fluorine is the most reactive element of all in Group 7.
What are the trends of group 7?
In group 7, the further down the group an element is, the higher its melting point and boiling point . This is because, going down group 7: the molecules become larger. more energy is needed to overcome these forces.
Why does reactivity increase down group 1 but up group 7?
Halogens from bromide to fluorine get more reactive because the force of attraction between the nucleus (core) and the outer electron get stronger as you go up group 7 elements. … As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive.
Why does reactivity decrease down?
The reactivity of Group 7 elements decreases down the group. … The electrons in the outer shell move further away from the nucleus as we go down the group and the attraction force between the electrons and the nucleus become weaker and weaker. This weaker attraction in the larger atoms makes it harder to gain electron.
What happens to reactivity up a group?
That is why as you go up a group Chemical Reactivity increases because it is easier for elements to gain electrons when they have high electronegativity. And the farther they are from the righ side of the table, the weaker their electronegativity is, resulting in lower Chemical Reactivity.
What is the trend for reactivity of metals across a period and the reason it occurs?
Across each period, from left to right, the increasing attraction between the nuclei and the outermost electrons causes the metallic character to decrease. Conversely, non-metallic character generally decreases down groups and increases across a period.
What means reactive?
To be reactive is to be ready to react or respond to something else — as opposed to ready to act on one’s own. A person who’s reactive does things only in response to others. … But if you’re a reactive person, then you only react; you’re always ready to react but not to act on your own.
Are metals reactive or nonreactive?
Metals tend to readily lose electrons and form cations. Most of them react with atmospheric oxygen to form metal oxides. However, different metals have different reactivities towards oxygen (unreactive metals such as gold and platinum do not readily form oxides when exposed to air).
How does reactivity of non metals vary down a group?
The reactivity of non – metals increases from left to right in a period whereas reactivity decreases in a group as we go down the group because the tendency to accept electrons decreases down the group.
Does reactivity of metals increase down a group?
Down the group, the effective nuclear charge experienced by valence electrons is decreasing because the outermost electrons are far away from the nucleus. Thus, these electrons can be lost easily by the element to form positive ions. Hence, the chemical reactivity of metals increases on going down a group.
How does reactivity change down group 1 and group 2 elements?
Reactivity increases down the group. This is because the smaller the atom the closer the outer electrons are to the nucleus. Therefore there is a greater attraction between the nucleus and electrons in magnesium than there is in calcium.
Is Group 13 reactive?
Moreover, all of the other elements in group 13 are relatively reactive at moderate temperatures, while boron’s reactivity only becomes comparable at very high temperatures. One characteristic that all do have in common is having three electrons in their valence shells.
Why does reactivity increase down Group 16?
The alkali metals lose electrons and reactivity increases as you go down the group. This is because the increased number of electron shells results in more shielding and a greater distance between the outer electrons and the nucleus, which reduces the attraction of the electrons to the nucleus.
What is the trend in reactivity of Group 2 elements with water?
The overall trend, for the reactivity of Group 2 metals with water, is an increase down the group. The outer electrons are easier to remove as they are further from the nucleus and there is more shielding resulting in a lower nuclear attraction.
What is the trend in reactivity of the halogens?
Unlike the group 1 metals, reactivity decreases as you go down the halogens. This means that fluorine, at the top of the group, is the most reactive.

