What is true about an ionic compound
They are made of metals and nonmetals is true of ionic compounds. They usually form between elements on opposite sides of the periodic table. Ionic compounds are only good conductors when molten or dissolved. When dissolved, they form ions, charged particles.
What describes an ionic compound?
Ionic compounds consist of oppositely charged ions that are held together by ionic bonds. The opposite charges cancel out so ionic compounds have a net neutral charge. Ionic compounds form when metals transfer valence electrons to nonmetals. … Ionic compounds are solids with high melting and boiling points.
Which describes ionic compounds quizlet?
Ionic compounds form crystals instead of molecules. Ionic bonds are strong and the crystals are rigid. As a result, ionic compounds are brittle solids with high melting and boiling points. In the liquid state or dissolved in water, ionic compounds are good conductors of electricity.
Which is true about ionic bond *?
Ionic bonding is the complete transfer of valence electron(s) between atoms. It is a type of chemical bond that generates two oppositely charged ions. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion.What is true for ionic bonding and ionic compounds?
Which is true of ionic compounds? They are made of metals and nonmetals is true of ionic compounds. They usually form between elements on opposite sides of the periodic table. Ionic compounds are only good conductors when molten or dissolved.
Which statement best describes how an ionic bond forms?
Which statement best describes how an ionic bond forms? The transfer of electrons forms strong bonds between ions. The sharing of electrons forms strong bonds between ions.
Which is true of all covalent compounds?
All covalent compounds share electrons in bonds. They are made of two or more nonmetals which share electrons. They do not form ions, charged particles, so they are not good conductors in water.
How are ionic compounds formed quizlet?
ionic bonds form when electrons are transferred from one atom to another atom. ions of different elements can combine by forming ionic bonds . positive ions & negative ions form when atom s lose or gain electrons.What is an ionic bond definition quizlet?
Ionic bonds is a bond that forms when electrons is being transferred from one form to another form. … Ionic bonds form so that the outermost energy level of atoms are filled. Ion. an atom or group of atoms that bring out a positive or negative electric charge as a result of having lost or gained one or more electrons.
What is formed by ionic bonds?Ionic bonds are formed between cations and anions. A cation is formed when a metal ion loses a valence electron while an anion is formed when a non-metal gains a valence electron. They both achieve a more stable electronic configuration through this exchange.
Article first time published onWhat is true about ionic and covalent compounds?
An ionic bond essentially donates an electron to the other atom participating in the bond, while electrons in a covalent bond are shared equally between the atoms. The only pure covalent bonds occur between identical atoms. … Ionic bonds form between a metal and a nonmetal. Covalent bonds form between two nonmetals.
What must generally be true for each ion in an ionic compound?
Because the ionic compound must be electrically neutral, it must have the same number of positive and negative charges. Two aluminum ions, each with a charge of 3+, would give us six positive charges, and three oxide ions, each with a charge of 2−, would give us six negative charges.
Which compound is an example of a binary ionic compound?
A binary ionic compound is composed of ions of two different elements – one of which is a metal, and the other a nonmetal. For example, iron(III) iodide, FeI3, is composed of iron ions, Fe3+ (elemental iron is a metal), and iodide ions, I- (elemental iodine is a nonmetal).
Which characteristic S makes ionic compounds useful as a building material?
Which characteristic(s) makes ionic compounds useful as a building material? Anna stated that ionic compounds have high melting points and low boiling points.
Which pair of atoms will form an ionic compound?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals.
How do ionic bonds affect the properties of ionic compounds?
How do ionic bonds affect the properties of ionic compounds? The bonds prevent electrons from moving throughout the crystal, so a solid ionic compound is a poor conductor. … One compound is covalent, and the other is ionic.
What is the relationship between molecules and compounds?
Molecules are groups of two or more atoms, but there is a special sub-group of molecules, too. These are compounds, and a compound is a group of two or more different atoms chemically bonded together. The key here is that compounds are made of at least two different elements.
How can you represent the composition of an ionic compound?
The composition of an ionic compound is represented with the help of ratio of cations to anions in the empirical formula where empirical formula can be defined as the simplest formula which provides the lowest whole number ratio of atoms which exist in the compound and it tells us about the relative number of every …
How do ionic compounds form crystals?
The ions have a regular, repeating arrangement called an ionic lattice . The lattice is formed because the ions attract each other and form a regular pattern with oppositely charged ions next to each other. … This is why solid ionic compounds form crystals with regular shapes.
Which statement is not true of ions?
When an element loses electrons it becomes a positively charged ion called a cation. Metals: tend to lose electrons to become positively charged ions (+) called cations. The statement that is not true about ions is d. When a cation forms, more electrons are transferred to it.
Which of the following is not true for ionic compounds?
Ionic compounds have high boiling point. Ionic compounds have low melting point. Ionic compounds are generally soluble in water. Ionic compounds conduct electricity in their molten state.
Do ionic compounds form molecules?
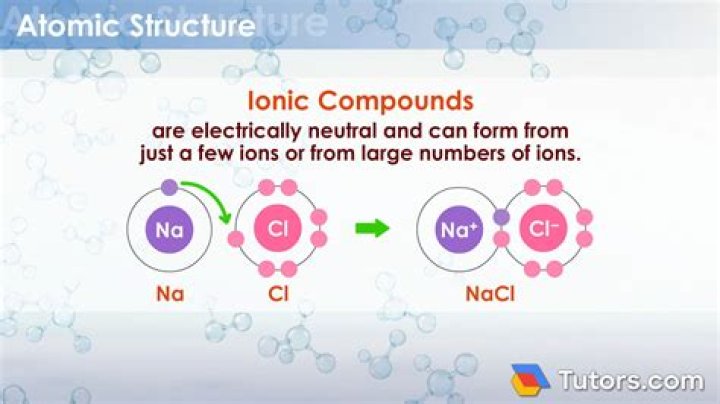
Compounds can be covalent or ionic. … The strong electrostatic attraction between adjacent cations and anions is known as an ionic bond. The most common example of an ionic compound is sodium chloride NaCl, better known as table salt. Unlike covalent compounds, there is no such thing as a molecule of an ionic compound.
Why are ionic compounds stable?
Ionic compounds are stable due to presence of strong electrostatic force of attraction between two ions.
Which of the statement is true about compound?
The true statement about compounds is- c) Compound is homogeneous and has a fixed composition. A compound is made up of two or more different elements that are evenly distributed throughout the compound. In a compound, the different elements are chemically combined in a definite proportion of their mass.
What is one thing that is true about ionic bonds but not covalent bonds?
Ionic bonds can form between a metal and a nonmetal, while covalent bonds can form between two nonmetals. … Ionic bonds are formed between two oppositely charged ions.
What is true about nonmetals in ionic bonds?
An ionic bond is formed between a metal and a non-metal. Non-metals(-ve ion) are “stronger” than the metal(+ve ion) and can get electrons very easily from the metal. These two opposite ions attract each other and form the ionic bond.
What is the overall charge of ionic compounds?
Ionic compounds contain positively and negatively charged ions in a ratio that results in an overall charge of zero.
Which set of elements is most likely to form an ionic compound?
A pair of elements will most likely form an ionic bond if one is a metal and one is a nonmetal. These types of ionic compounds are composed of monatomic cations and anions.
How do you know if a compound is molecular or ionic?
As a general rule of thumb, compounds that involve a metal binding with either a non-metal or a semi-metal will display ionic bonding. Compounds that are composed of only non-metals or semi-metals with non-metals will display covalent bonding and will be classified as molecular compounds.
What makes a binary compound ionic?
A binary ionic compound is composed of ions of two different elements – one of which is a metal, and the other a nonmetal. For example, sodium iodide, NaI, is composed of sodium ions, Na+ (elemental sodium is a metal), and iodide ions, I- (elemental iodine is a nonmetal).
What is meaning of binary compound?
Binary compounds are chemical compounds comprising of two distinct elements. An element is a substance that cannot be further divided into any simpler substances using chemical methods. Hence sodium fluoride, magnesium oxide, and calcium chloride are all chemical elements.

