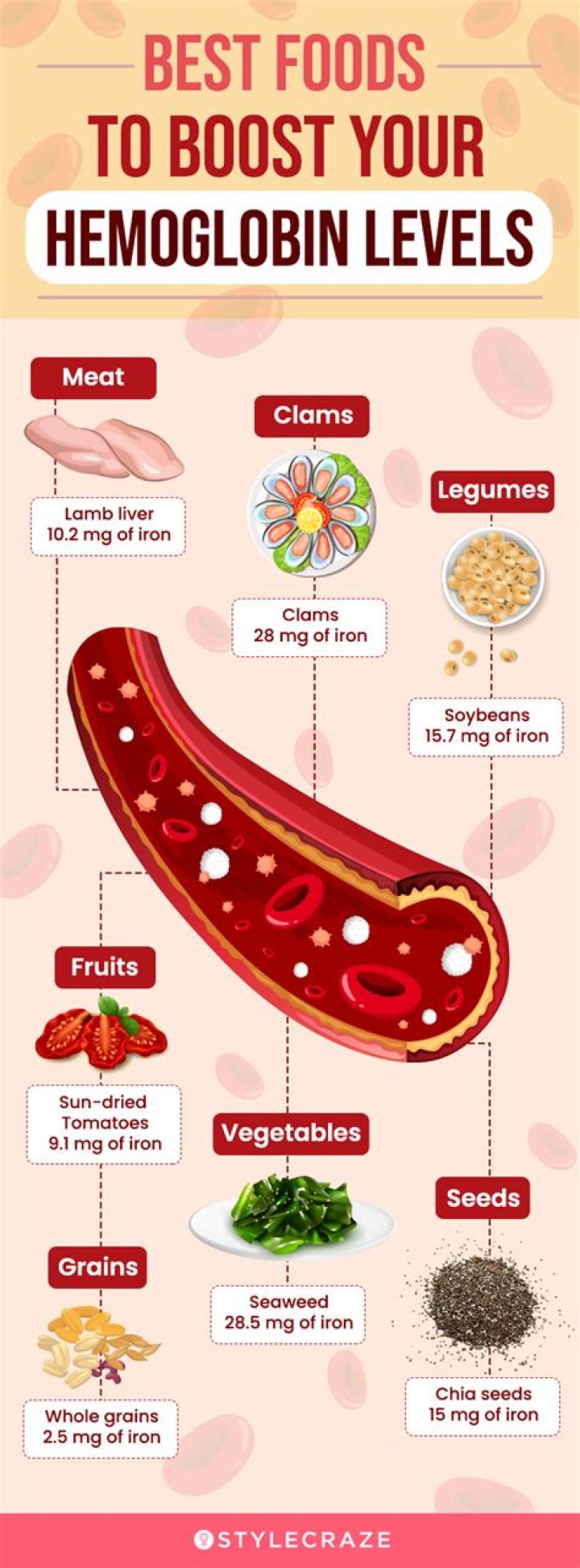
What percentage of water is found in CuSO4 * 5h2o
A mole of CuSO4•5H2O contains 5 moles of water (which corresponds to 90 grams of water) as part of its structure. Therefore, the substance CuSO4•5H2O always consists of 90/250 or 36% water by weight. The water is a definite part of the structure of the compound, and the substance should not be considered to be “wet.”
What is the percentage of copper in CuSO4 * 5H2O?
Determine the concentration by mass percent by dividing the formula weight of CuSO4 by the formula weight of CuSO4-5H2O and multiplying by 100 percent: 159.62 / 249.72 * 100 = 63.92 percent. This means that a 100-gram sample of copper sulfate pentahydrate will contain 63.92 grams of copper sulfate.
What percentage of water h2o is found in the hydrate na2s ● 9h2o?
Sodium sulfate decahydrate is 55.9%H2O .
What hydrate is CuSO4 5H2O?
Copper sulfate pentahydrate. Copper(II) sulfate pentahydrate is the pentahydrate of copper(2+) sulfate. A bright blue crystalline solid. It is a hydrate and a metal sulfate.What is the color of CuSO4 * 5H2O?
CuSO4· 5H2O is blue in colour whereas anhydrous CuSO4 is colourless.
How do you calculate percent by mass?
To calculate the mass percent of an element in a compound, we divide the mass of the element in 1 mole of the compound by the compound’s molar mass and multiply the result by 100.
What percent of CuSO4 is copper?
Commercial copper sulfate is usually about 98% pure copper sulfate, and may contain traces of water. Anhydrous copper sulfate is 39.81 percent copper and 60.19 percent sulfate by mass, and in its blue, hydrous form, it is 25.47% copper, 38.47% sulfate (12.82% sulfur) and 36.06% water by mass.
What is the percentage composition of water?
The percentage of an element in a compound is 100 times the fraction, so for water the mass percent hydrogen is 11.11% and the mass percent oxygen is 88.89%.What is the percentage composition of na2s 23.5 h2o?
ElementSymbolMass PercentSodiumNa27.349%HydrogenH5.995%OxygenO47.583%SulfurS19.073%
What is the percent by mass of water in the hydrate na2so4 10h2o GFM of the anhydrous na2so4 is 142 g mol?The percent by mass of water in Na2 SO4 x 10H2 O is 55.91%.
Article first time published onWhat is the colour of 5H2O?
5H2O is blue . The colour of any vol. of water or 5H2O is colourless .
What is the colour of CuSO4 without water?
In hydrated CuSO4 the water molecules surrounding the Central metal (Cu) function as ligands which bring d-d transition and hence emits blue colour in visible region due which hydrated CuSO4 appears blue and as anhydrous CuSO4 doesn’t have any water of crystallisation hence remains white in colour.
What is the difference between CuSO4 and CuSO4 5H2O?
The key difference between CuSO4 and CuSO4 5H2O is that CuSO4 is amorphous, whereas CuSO4 5H2O is crystalline. CuSO4 is the chemical formula of copper(II) sulfate, while CuSO4 5H2O is the hydrated form of copper(II) sulfate.
What type of reaction is CuSO4 5H2O?
ReactionBalanced Chemical ReactionType of Chemical ReactionCuSO4(s) + H2O(l) → CuSO4*5H2O(s)CuSO4(s) + 5H2O(l) → CuSO4*5H2O(s)SynthesisCa(s) + S(s) → CaS(s)Ca(s) + S(s) → CaS(s)SynthesisMg(s) + O2(g) → MgO(s)2Mg(s) + O2(g) → 2MgO(s)Synthesis
How do you find the mass percent of water in a hydrate?
Divide the mass of the water lost by the mass of hydrate and multiply by 100. The theoretical (actual) percent hydration (percent water) can be calculated from the formula of the hydrate by dividing the mass of water in one mole of the hydrate by the molar mass of the hydrate and multiplying by 100.
How can I calculate percentage?
Percentage can be calculated by dividing the value by the total value, and then multiplying the result by 100. The formula used to calculate percentage is: (value/total value)×100%.
What is percentage by volume?
Volume/Volume Percentage Definition Volume/volume percentage (v/v percent) is a measure of the concentration of a substance in a solution. It is expressed as the ratio of the volume of the solute to the total volume of the solution multiplied by 100.
How many moles of water are in 0.250 mol of CuSO4 5H2O?
How many moles of water are contained in 0.250 moles of CuSO4*5H2O? Ratio of CuSO4*5H2O is 1:5. One mole of CuSO4 is 1 CuSO4*5H2O ratio. 0.250 moles of CuSO4*5H2O is 0.250 moles of CuSO4 (0.250×1) and 1.25 moles of H2O (0.250×5=1.25).
What is the empirical formula for CuSO4 hydrate?
The value x = 5.0755 can be approximate to x = 5; therefore, the empirical formula is CuSO4· 5H2O. As stated by theory, when heating the crystals, the water will evaporate depending on the temperature. Repeating heating and stirring the crystals can ensure the hydrated CuSO4 is dehydrated completely.
What happens when anhydrous copper sulphate is added to water?
Water being added to some anhydrous copper (II) sulphate. The anhydrous form is pale green in colour. When water is added slowly, the copper sulphate becomes hydrated, with five water molecules binding to each copper sulphate molecule. This pentahydrate form is blue.
What is the percentage of calcium in calcium carbonate?
Therefore, the percentage of calcium in calcium carbonate is 40.04%.
What is the percentage composition of Na2S2O3?
ElementSymbolMass PercentSodiumNa29.081%OxygenO30.358%SulfurS40.561%
What is the symbol for sodium?
sodium (Na), chemical element of the alkali metal group (Group 1 [Ia]) of the periodic table. Sodium is a very soft silvery-white metal. Sodium is the most common alkali metal and the sixth most abundant element on Earth, comprising 2.8 percent of Earth’s crust.
How do you find the percent composition?
- Find the molar mass of all the elements in the compound in grams per mole.
- Find the molecular mass of the entire compound.
- Divide the component’s molar mass by the entire molecular mass.
- You will now have a number between 0 and 1. Multiply it by 100% to get percent composition.
What is the percent composition of C6H12O6?
Percent Composition An elemental analysis of a white crystalline compound believed to be C6H12O6, analyzed for C, H and O gave the following results. C = 40.00 % , H = 6.71 %, O = 53.28 % by wt.
How do you find the mass percent of each element in water?
In chemistry, finding the mass percentage of an element in a compound might sound complicated, but the calculation is simple. For example, to determine the mass percentage of hydrogen in water (H2O), divide the molar mass of hydrogen by the total molar mass of water and then multiply the result by 100.
What is the percentage composition of na2so4?
We may say sodium sulfate is 32.37% sodium, 22.57% sulfur, and 45.06% oxygen; or, 32.37% sodium and 67.63% sulfate.
Why is CuSO4 5h20 blue?
It is so because of the presence of water of crystallisation which facilitates the transition which transmits color to the compound. Complete step by step answer: The hydrated copper sulphate \[CuS{O_4} \cdot 5{H_2}O\] is blue in color. The ligand (water molecules) cause splitting of d – orbitals.
Why CuSO4 5H2O is blue in colour while CuSO4 is Colourless?
In CuSO4. 5H2O water acts as ligand as a result it causes crystal field splitting. Hence d—d transition is possible in CuSO4. … In the anhydrous CuSO4 due to the absence of water ligand crystal field splitting is not possible and hence no colour.
What is colour of CuSO4?
The colour of hydrated copper sulphate is blue.
What happens when CuSO4 5H2O is heated?
If copper sulphate 5 – water is heated, anhydrous copper sulphate is formed.