Which diene is more reactive
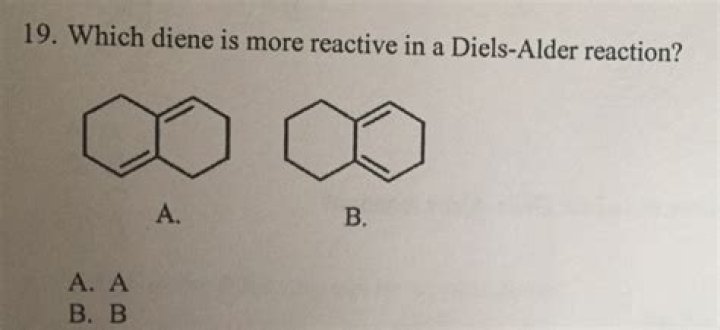
The most reactive dienophile is the aldehyde — propenal.
How do you rank up diene reactivity?
Dienophile reactivity is increased by: (A) electron-withdrawing substituents, (B) a weak π-bond, and (C) release of ring strain.
Which Diels-Alder reaction happens fastest?
In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups) and electron-withdrawing groups on the dienophile.
How do you know which diene is more stable?
Conjugated dienes are more stable than non conjugated dienes (both isolated and cumulated) due to factors such as delocalization of charge through resonance and hybridization energy. This can also explain why allylic radicals are much more stable than secondary or even tertiary carbocations.Is conjugated diene more stable than alkene?
Conjugated dienes are more stable than non conjugated dienes (both isolated and cumulated) due to factors such as delocalization of charge through resonance and hybridization energy. … Along with resonance, hybridization energy effect the stability of the compound.
Which Dienes will react with ethene in a Diels-Alder reaction?
Since Diels-Alder reactions are highly stereospecific, ethene will only react with s-cis conformation dienes.
Is anthracene a diene?
Anthracene acts as the diene and maleic anhydride functions as the dienophile. Xylene (dimethylbenzene) is used as a high boiling temperature solvent so that the reaction will proceed quickly .
What is an S cis diene?
S-cis: The conformation of a single bond separating two double bonds in which the double bonds have a dihedral angle of 0o (the double bonds lie in the same plane and point in the same direction). The ‘s’ in s-cis stands for ‘single’.What are the 3 types of dienes?
- Cumulated dienes have the double bonds sharing a common atom. …
- Conjugated dienes have conjugated double bonds separated by one single bond. …
- Unconjugated dienes have the double bonds separated by two or more single bonds.
Correct Option- i Conjugated alkadiene CH2 =CH—CH =CH2 Explanation-i Due to resonance conjugated alkadiene is maximum stable. Isolated is more stable than cumulated alkadiene due to H-effect .
Article first time published onWhat makes a diene reactive?
Remember that electron-donating groups increase the reactivity of the diene: … Therefore, electron-donating groups on the diene increase its reactivity, while electron-withdrawing groups on the dienophile lower the LUMO energy level, thus support this electron flow as well.
Is Diels-Alder endo or exo?
Long story short: the Diels-Alder is another example of a reaction that can be run under kinetic or thermodynamic control, where the “endo” is the kinetic product and the “exo” is the thermodynamic product. ]
Is Endo or Exo The major product?
When Diels and Alder originally discovered this phenomenon, they assigned the name “endo” to the major product (where the dienophile is pointing “in”, towards the alkene) , and the term “exo” (outside, such as in “exoskeleton”) to refer to the minor product (where the dienophile is pointing “out”, away from the alkene) …
Are isolated or cumulated dienes more stable?
Conjugated dienes are characterized by alternating carbon-carbon double bonds separated by carbon-carbon single bonds. Cumulated dienes are characterized by adjacent carbon-carbon double bonds. While conjugated dienes are energetically more stable than isolated double bonds. Cumulated double bonds are unstable.
Which of the following is conjugated diene?
2,4-Hexadiene is a conjugated diene: The structures of the compounds are given below.
What is difference between alkene and diene?
Dienes are alkenes with 2 double bonds. … Compounds containing two carbon-carbon cumulated double bonds are called allenes. Conjugated dienes differ from simple alkenes in that they. are more stable, undergo 1,4-addition, and are more reactive.
Is anthracene a diene or dienophile?
Anthracene acts as the diene and maleic anhydride functions as the dienophile. Xylene (dimethylbenzene) is used as a high boiling temperature solvent so that the reaction will proceed quickly.
Why is anthracene a diene?
Anthracene, however, is an unusually unreactive diene. This is due to both steric effects, but more importantly because the “diene” is really part of an aromatic ring system and is thus stabilized. This stabilization in the reactant reduces the reactivity (stability/reactivity principle).
What are Dienes and Dienophiles?
As nouns the difference between diene and dienophile is that diene is (organic chemistry) an organic compound, especially a hydrocarbon, containing two double bonds while dienophile is (organic chemistry) a compound that readily reacts with a diene; especially an alkene in the diels-alder reaction.
Which of the following is NOT type of dienes?
We know that cumulated diene has two continuous double bonds C=C=C structure. Therefore only penta-1, 3-diene is not a cumulated diene. It does not have continuous double bonds around the carbon atoms. Hence option ‘C’ is the correct answer.
Why is a cumulated diene less stable?
Cumulated dienes are typically less stable than other alkenes. The main reason for the instability is the fact that this sort of diene is a probable transition state for an alkyne’s triple bond to move down the carbon chain towards the most stable position.
What are dienes give example?
Dienes are compounds whose molecules contain two carbon-carbon double bonds separated by a single bond. The most important diene polymers—polybutadiene, polychloroprene, and polyisoprene—are elastomers that are made into vulcanized rubber products.
What is the cis configuration?
cis configuration: configuration of a geometrical isomer in which two groups are on the same side of an imaginary reference line on the molecule.
What is a conjugated triene?
A conjugated triene contains three alternating double bonds. … An unconjugated diene has the two double bonds of the molecule separated by more than one single bond. 2. Pi electrons cannot be delocalized in unconjugated systems.
Which of the following is a cumulated diene?
Allylene. H2C=C=CH2 (Allene) Allene is an example of cummulated diene.
Which of the following is most stable compound?
Triphenyl methyl cation has three benzene resonating ring so it is most stable compound.
What are cumulated dienes give example?
The 1,2-dienes, which have cumulated double bonds, commonly are called allenes. The simplest example is 1,2-propadiene, … In this formulation each of two electrons of the central atom form collinear spσ bonds to the terminal sp2-hybridized carbons.
Which diene reacts more rapidly in Diels Alder reactions cyclopentadiene or 1/3 butadiene briefly explain your choice?
The cyclopentadiene will react more quickly. The reason why the cyclopentadiene reacts more quickly is because it has a fixed conformation….
Why is cyclopentadiene reactive?
Cyclopentadiene is highly reactive in Diels–Alder reactions because only minimal out-of-plane distortion is required to achieve the transition state geometry compared with that of other cyclic and acyclic dienes. Asynchronous transition states have significant out-of-plane distortion about only one double bond.
Why is exo more stable?
The exo product is most stable as the smaller one-atom bridge eclipses the anhydride ring causing less steric hindrance, and is the thermodynamic product. Under reversible conditions, the exo product is formed.
How do you know if its exo or endothermic?
So if the sum of the enthalpies of the reactants is greater than the products, the reaction will be exothermic. If the products side has a larger enthalpy, the reaction is endothermic.

