Why are the emission wavelengths for helium and hydrogen different
And, the key difference between hydrogen and helium emission spectra is that the helium emission spectrum has more lines than that of the hydrogen emission spectrum. It is mainly because hydrogen has one electron per atom while helium has two electrons per atom.
Why do helium and hydrogen have different emission wavelengths?
Unlike in hydrogen, there are electron-electron repulsions and different nuclei-electron attractions in the helium atom. Therefore, different spectra (different from hydrogen) comes out with different wavelengths for the helium atom.
Why is the absorption spectrum of atomic hydrogen different from that of helium?
Hydrogen, with one proton in the nucleus, has a different field configuration than does Helium with two protons – this is why the two atoms have a different energy levels and different characteristic absorption and emission lines.
Why does hydrogen have less emission lines?
Those photons appear as lines. For this reason, though hydrogen has only one electron, more than one emission line is observed in its spectrum. This arises due to the electron making transitions between the various energy levels in an atom .Why do the hydrogen and neon emission lamps have different spectra?
For atoms with more than one electron, the physics becomes much more complex, but the basic idea that electrons have only certain allowed orbits still holds. Each element has a different set of allowed orbits, so each element emits or absorbs photons with different energies — and therefore, different wavelengths.
What is the emission spectrum of helium?
The 12 lines of the visible helium spectrum correspond to wavelengths of 388.8, 447.1, 471.3, 492.1, 501.5, 504.7, 587.5, 667.8, 686.7, 706.5, 728.1 and 781.3 nanometres (nm).
Does helium have more energy than hydrogen?
Helium has a structure 1s2. The electron is being removed from the same orbital as in hydrogen’s case. It is close to the nucleus and unscreened. The value of the ionization energy (2370 kJ mol-1) is much higher than hydrogen, because the nucleus now has 2 protons attracting the electrons instead of 1.
Why are there more emission lines than absorption?
In the emission spectrum, the electrons in the energy levels usually start at random energy levels and so there is more of a variety of wavelengths that could possibly be emitted.Why does hydrogen only have 4 emission lines?
This is explained in the Bohr model by the realization that the electron orbits are not equally spaced. … The electron energy level diagram for the hydrogen atom. He found that the four visible spectral lines corresponded to transitions from higher energy levels down to the second energy level (n = 2).
Why do different elements produce different spectral lines?Each element has its own unique line spectrum and is thus referred to as the “fingerprint” for a particular element. The spectra for each element are unique because each element contains differing numbers of electrons and thus different energy levels.
Article first time published onWhy do the wavelengths of the absorption and emission lines match?
Because the energy levels in an element’s atoms are fixed, the size of the outward jumps made by the electrons are the same as the inward jumps. Therefore, the pattern of absorption lines is the same as the pattern of emission lines.
Why is the absorption spectrum of atomic hydrogen different from that of helium quizlet?
Why is the absorption spectrum of atomic hydrogen different from that of helium? The energy differences between orbitals in hydrogen are different from those in helium. … When an atom emits light, an electron in it moves from a higher to a lower energy level.
What is difference between emission spectrum and absorption spectrum?
The main difference between emission and absorption spectra is that an emission spectrum has different coloured lines in the spectrum, whereas an absorption spectrum has dark-coloured lines in the spectrum.
Why does each element have a unique gas phase emission spectrum?
(d) Each element displays a unique gas-phase emission spectrum. Each element has a unique set of quantized energy states for its electrons (because of its unique nuclear charge and unique electron configuration).
Why the emission spectrum of hydrogen atom is a line spectrum?
The emission spectrum of atomic hydrogen has been divided into a number of spectral series, with wavelengths given by the Rydberg formula. These observed spectral lines are due to the electron making transitions between two energy levels in an atom.
Why is the emission spectrum of hydrogen a line spectrum?
Lines in the spectrum were due to transitions in which an electron moved from a higher-energy orbit with a larger radius to a lower-energy orbit with smaller radius. The orbit closest to the nucleus represented the ground state of the atom and was most stable; orbits farther away were higher-energy excited states.
Why is helium ionization energy than hydrogen?
The value for helium is higher than that for hydrogen because there are now two protons in the nucleus. The nuclear charge is greater so the pull on the outer electrons is larger. More energy will be needed to pull an electron out of the atom.
Why is the ionisation energy of helium not four times that of hydrogen?
The first ionization energy for helium is slightly less than twice the ionization energy for hydrogen because each electron in helium feels the attractive force of two protons, instead of one. It takes far less energy, however, to remove an electron from a lithium atom, which has three protons in its nucleus.
Why is helium ionization energy so high?
Yes, Helium has the highest ionization energy! This is because the electrons in helium are very close to the nucleus and so the electrostatic attraction is very high. This makes it difficult to remove an electron.
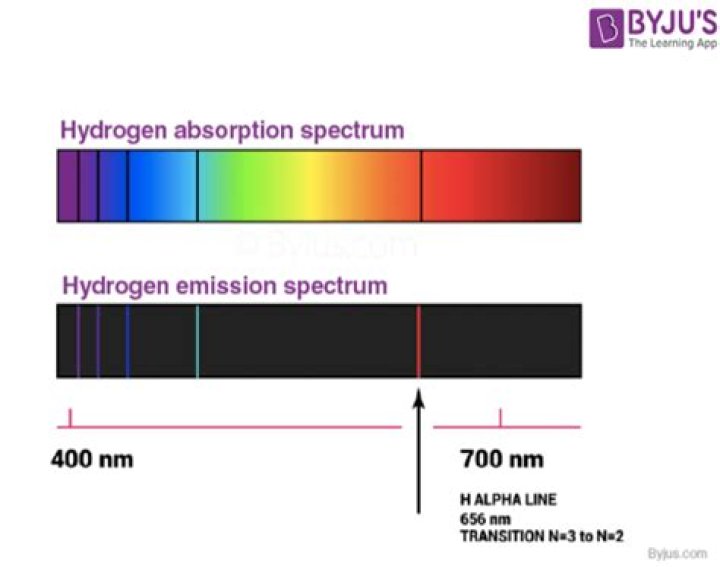
What wavelengths are found in the hydrogen spectrum?
The visible spectrum of light from hydrogen displays four wavelengths, 410 nm, 434 nm, 486 nm, and 656 nm, that correspond to emissions of photons by electrons in excited states transitioning to the quantum level described by the principal quantum number n equals 2.
What is the wavelength of the strongest helium absorption line?
Spectral LinesWavelengths (Angstroms)Titanium Oxidelots of lines from 4900 – 5200, 5400 – 5700, 6200 – 6300, 6700 – 6900G Band4250Sodium5800Helium (neutral)4200
Which spectrum is more complicated hydrogen or helium?
You might also notice that hydrogen is relatively simple, with few lines. Helium is a bit more complicated, and the elements below get increasingly complicated. Hydrogen spectrum is simple because there is only one electron in a hydrogen atom.
How many emission lines are possible for hydrogen?
So, there are four lines in the visible spectrum of hydrogen. There are, of course, lots of other lines in hydrogen’s spectrum but they are all outside of our visible range so we cannot see them with our naked eye (and a diffraction grating).
Why does neon have more spectral lines than hydrogen?
Neon has ten electrons. With ten electrons there are many more possible transitions of electrons from higher to lower energy levels and many more lines in the emission spectrum. Hydrogen only has one electron.
How can a hydrogen atom which has only one electron have so many spectral lines quizlet?
How can a hydrogen atom, which has only one electron, have so many spectral lines? The many spectral lines from the element hydrogen are the result of the many energy states the single electron can occupy when excited.
Are emission and absorption same?
Emission is the process of elements releasing different photons of color as their atoms return to their lower energy levels. Atoms emit light when they are heated or excited at high energy levels. … Absorption occurs when electrons absorb photons which causes them to gain energy and jump to higher energy levels.
What is happening within the hydrogen atom to create emission lines?
When electrons move from a higher energy level to a lower one, photons are emitted, and an emission line can be seen in the spectrum. Absorption lines are seen when electrons absorb photons and move to higher energy levels.
Why is light of different energy absorbed by different elements?
Explanation: The electrons in an atom can only occupy certain allowed energy levels. … Conversely, an atomic electron can be promoted to a higher energy level when it absorbs a photon. Again because only certain transitions are allowed, only certain wavelengths can be absorbed.
What is the wavelength of the strongest hydrogen absorption line?
Spectral LinesWavelengths (Angstroms)Hydrogen: Ha, Hb, Hg6600, 4800, 4350Ionized Calcium: H and K Lines3800 – 4000Titanium Oxidelots of lines from 4900 – 5200, 5400 – 5700, 6200 – 6300, 6700 – 6900G Band4250
Why are some emission lines brighter than others?
In hydrogen spectrum,some spectral lines are brighter than others depending upon their energy level. … When electron jumps from some higher orbit,the energy released in the from of photon will be greater,and we get a brighter line. Thus in hydrogen spectrum some lines are brighter than others.
What is the connection between the absorption and emission spectrum?
The difference between absorption and emission spectra are that absorption lines are where light has been absorbed by the atom thus you see a dip in the spectrum whereas emission spectra have spikes in the spectra due to atoms releasing photons at those wavelengths.