Why does NaCl dissolve in water even though it is endothermic
So yes, dissolving is an endothermic process because the water is gaining energy from the NaCl ionic bond or lattice, thus from the Gibbs free energy equation, G must be negative and that free energy is mostly controlled by the value of the entropy; not the enthalpy in the system.
Why is NaCl dissolving in water endothermic?
Dissolution of sodium chloride (table salt) in water is endothermic. … This is because more energy is released upon formation of solute-solvent bonds than was required to break apart the hydrogen bonds in water, as well as the ionic bonds in KOH.
Is the reaction of NaCl endothermic or exothermic?
When NaCl forms, what really happens is that Na+ and Cl− come together and form an ionic bond and thus NaCl is formed and it is a quite exothermic reaction.
Why does NaCl dissolve in water?
When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. … Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together.Is the dissolution of NaCl S in water endothermic or exothermic?
The dissolution of sodium chloride in water is SLIGHTLY endothermic.
Is sodium nitrate endothermic or exothermic?
It is exothermic. The heat is released to the surroundings which is the water making the solution.
Is dissolving MgSO4 in water exothermic or endothermic?
Magnesium sulfate is an inorganic salt with the chemical formula MgSO4. It is highly soluble in water and its dissolution is an exothermic reaction: heat is given off.
Why does NaCl dissolve in h2o despite its strong ionic bond?
The reason salt dissolves in water is therefore due to how, the positively charged sodium ions are attracted to the negative polar area of the water molecule. Similarly, the negatively charged chloride ions are attracted to the positive polar area of the water molecule.Why does NaCl dissolve in water quizlet?
Water dissolves in salt because the negative part of a water molecule, the oxygen part is attracted to the positive sodium ion of the salt. Also, the positive part of the water molecule, the hydrogen part is attracted to the negative chloride ion of the salt.
Why is dissolving salts sometimes an endothermic process and sometimes an exothermic process?It takes just slightly more energy to separate the ions from one another than is released from the water molecules surrounding the ions. This means just slightly more energy must be put into the solution than is released back into the solution; therefore dissolving table salt in water is endothermic.
Article first time published onWhy is NaCl an exothermic reaction?
NaCl exists as a crystal lattice of sodium and chlorine ions, where the extra electron from a sodium atom fills in the “hole” needed to complete a chlorine atom’s outer electron shell. … While the gain of an electron by a nonmetal is usually exothermic (energy is released when the nonmetal gains a full octet).
Why is dissolution exothermic?
The process of dissolving is exothermic when more energy is released when water molecules “bond” to the solute than is used to pull the solute apart. Because more energy is released than is used, the molecules of the solution move faster, making the temperature increase. Project the image Endothermic Dissolving.
Is endothermic positive or negative?
So, if a reaction releases more energy than it absorbs, the reaction is exothermic and enthalpy will be negative. Think of this as an amount of heat leaving (or being subtracted from) the reaction. If a reaction absorbs or uses more energy than it releases, the reaction is endothermic, and enthalpy will be positive.
How do you know if a solution is exothermic or endothermic?
So if the sum of the enthalpies of the reactants is greater than the products, the reaction will be exothermic. If the products side has a larger enthalpy, the reaction is endothermic.
Why is MgSO4 endothermic?
Its all a matter of breaking bonds (endothermic) and making bonds (exothermic). It requires energy (heat) to break up the magnesium sulphate lattice (solute) into individual ions, so that process is endothermic.
What happens when MgSO4 dissolves in water?
When dissolved in water, magnesium sulfate ionizes (separates) into magnesium (Mg2+) ions and sulfate (SO42-) ions. Solutions of magnesium sulfate have a neutral pH. Magnesium sulfate is used in many industrial processes and in the manufacturing of fertilizers.
What happens when MgSO4 reacts with water?
It can also be prepared commercially by the reaction of magnesium carbonate (MgCO3) with sulfuric acid (H2SO4). … When dissolved in water, magnesium sulfate ionizes (or separates into ions) into magnesium (Mg2+) ions, and sulfate (SO 2- 4 ) ions. Solutions of magnesium sulfate have a neutral pH.
Why does NaNO3 dissolve in water?
NaNO3 is ionic. Very ionic. Both Na+ and NO3- are stable in ionic form, and the solid crystal is held together by strong electrostatic charges rather than any electron sharing. Water, as a polar molecule, surrounds each ion allowing it to dissociate from the ionic crystal and keeps it separated (dissolves it).
Is dissolving NaNO3 in water endothermic or exothermic?
Ammonium nitrate dissolving in solution is an endothermic reaction. As the ammonium nitrate dissolves, heat energy is absorbed from the environment causing the surrounding environment to feel cold.
When salts are dissolved in water they quizlet?
When salt is placed in water the atoms of the salt start to separate from each other making the molecule rip apart because salt has a negative and a positive side making it polar and water also is polar and polar molecules attract to other polar molecules.
What is the name of the process that occurs when NaCl is placed in water?
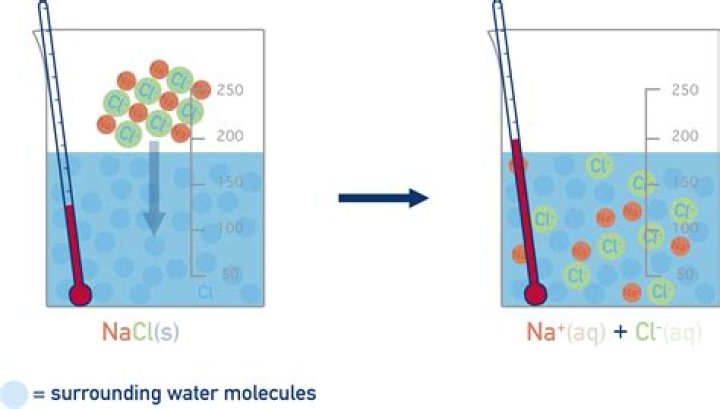
1: The dissolving of sodium chloride in water. After coming apart from the crystal, the individual ions are then surrounded by solvent particles in a process called solvation. Note that the individual Na+ ions are surrounded by water molecules with the oxygen atom oriented near the positive ion.
When salt dissolves completely into water which term is used to describe the water?
Salt dissolved in water is a solution. The major component of a solution, called the solvent, is typically the same phase as the solution itself. Each minor component of a solution (and there may be more than one) is called the solute.
Is salt and water exothermic or endothermic?
Dissolving salt in water is endothermic. This means that when salt is dissolved in water the temperature of the solution is often a little lower than the temperature of the water in which the salt is dissolved.
Why is sodium hydroxide dissolution exothermic?
Dissolving sodium hydroxide in water is exothermic. This is because the sodium ions and hydroxyl ions are capable of interacting with the water…
Is the dissolution of Naoh endothermic or exothermic?
Similar to the hydration of sulfuric acid, dissolution of solid sodium hydroxide in water is a highly exothermic reaction where a large amount of heat is liberated, posing a threat to safety through the possibility of splashing. The resulting solution is usually colorless and odorless.
Why is NaCl a solid?
NaCl is an ionic compound and form strong inter ionic bonds among themselves to form a crystal structure, that is why NaCl is solid.
Is water freezing endothermic or exothermic?
When water becomes a solid, it releases heat, warming up its surroundings. This makes freezing an exothermic reaction.
What happens when NaCl is formed?
Sodium chloride is formed when sodium atoms interact with chlorine atoms. When this occurs, sodium will donate an electron (which is a negatively-charged particle) to chlorine. This makes sodium slightly positive and chlorine slightly negative.
What happens when NaOH dissolves in water?
When NaOH dissolves into water, it dissociates into two ions: a positively charged sodium ion and a negatively charged hydroxide ion (OH-). The increased number of hydroxide ions in the solution increases the concentration of hydroxide ions in the water.
Why is melting solid salts endothermic?
3. Melting ice is an endothermic process in which thermal energy from the environment causes the molecules of solid water to vibrate fast enough to change to a liquid. … Mixing water and potassium chloride, a salt, results in a cooler solution.
Why does salt dissolve in water but not oil?
Finally, salt doesn’t dissolve in oil at all because oil has practically no charge at all. Some of these relationships are shown in Figure 3. A difference in charge also explains why oil and water will not mix. Since oil molecules are almost entirely uncharged, they won’t mix with charged water molecules.

