Why was Sulphuric acid added in making up the ammonium iron sulphate solution
Sulphuric acid is added to prevent hydrolysis and to provide surplus H+ ions in the solution to keep reaction proceeding and also sulphuric acid is stable towards oxidation.
Why sulphuric acid is added while preparing standard ferrous ammonium sulphate?
Dilute sulphuric acid is added to prevent the hydrolysis of ferrous sulphate. For dissolving the mixture of salts in water excessive heating is avoided. This is for the prevention of Fe2+ ions (light green) to Fe3+ ions (yellow).
Why do we add sulphuric acid to Mohr's salt?
Ferrous ions of Mohr’s salt undergo hydrolysis in aqueous solution. To prevent the hydrolysis, Conc. H2SO4 needs to be added to the Mohr’s salt crystals during the preparation of its standard solution. Another reason is also to prevent the oxidation of Fe2+ ions of Mohr’s salt to Fe3+ ions.
Why is the solution of ammonium iron II sulfate acidified with sulfuric acid before titration?
Thus, during titrations, sulfuric acid is commonly added to solutions of Mohr’s salt to reduce air oxidation to ferric iron, thus, the oxidation would take place only in the presence of permanganate solution.Why is sulphuric acid used in redox titration?
Sulphuric Acid (H2SO4) is used in the redox titration process because it provides the H(+) ions necessary for the reaction to occur more quickly whilst the sulphate(-) ions barely react during the reaction. … Therefore, sulfuric acid is added to make the solution acidic.
Why it is necessary to add few drops of concentrated H2SO4 in the preparation of FAS solution?
The reason why you add sulfuric acid before adding water is that the oxidation of the ferrous ion by dissolved oxygen in water is fast at neutral to high pH. By dissolving FAS in water with no acid added and watching the solution turn a cloudy orange, indicating the formation of ferric Ions.
Why is H2SO4 added to FAS K2Cr2O7 titration?
Just use H2SO4, since it can’t be oxidised further and is thus safe from K2Cr2O7. It’s because chloride ions can be oxidised by strong oxidising agents such as permanganate (but also some others) in a reaction that will produce chlorine: .
Why is sulphuric acid added twice in FAS?
The reason why you add sulfuric acid before water is that the oxidation of the ferrous ion by dissolved oxygen in water is fast at neutral to high pH. You can see this yourself by dissolving FAS in water with no acid added and watching the solution turn a cloudy orange, indicating the formation of ferric ions.What is the purpose of adding the sulfuric acid?
Concentrated sulfuric acid is used as a catalyst, and has a dual role: Speeds up the reaction. Acts as a dehydrating agent, forcing the equilibrium to the right and resulting in a greater yield of ester.
Why is sulfuric acid added to the solutions?Sulphuric acid is added to prevent hydrolysis and to provide surplus H+ ions in the solution to keep reaction proceeding and also sulphuric acid is stable towards oxidation.
Article first time published onWhy h2so4 is added in kmno4 titration?
Titrations with Permanganate must be carried out in strong acid solution. Sulfuric Acid is generally used for this purpose because Nitric Acid and Hydrochloric Acid can participate in competing oxidation-reduction reactions, reducing the accuracy of the titration.
Why it is necessary to use both the sulfuric acid as well as phosphoric acid in the experiment?
Both sulfuric acid and phosphoric acid have multiple equilibrium constants, corresponding to each of the hydrogen ions that can dissociate. Acids with large equilibrium constants are strong acids. Sulfuric acid has a first equilibrium constant of 1.0 x 10^3, which makes it a strong acid.
Why is sulfuric acid added to the iron II solution prior to titration could hydrochloric acid or nitric acid be used instead of sulfuric acid explain?
Nitric acid and hydrochloric acid are not suitable for use instead of sulfuric acid in this experiment. The nitric acid is itself an oxidising agent, and the hydrochloric acid reacts with potassium manganate(VII) solution, being oxidised itself in the reaction.
Why do we add dilute sulphuric acid into water during electrolysis?
Because adding dilute sulphuric acid makes it slightly acidic, that mesns the water gets impure and thus electricity flows through it easily. In other words, adding sulphuric acid makes water more conductive thus making current flow through it.
What is the purpose of adding phosphoric acid in the Fe titration?
In the redox titration of iron(III) with permanganate or dichromate, we use phosphoric(V) acid to “mask” the color of iron(III) because it interferes with the end point color change.
Why do we use H2SO4 instead of HNO3 or HCl?
HCl isn’t suitable, as will be oxidised to chlorine gas by the permanganate whilst HNO3 is an oxidising agent itself and would interfere with the reaction. H2SO4 (as long as it’s dilute) doesn’t have this problem, so is the preferred medium for such oxidations (especially for titrations).
Why is it necessary to add few drops of conc?
It is less readily oxidised than FeSO4 and therefore, a better volumetric reagent in preference to ferrous sulfate. The addition of sulfuric acid in this experiment prevents the hydrolysis of this salt. Ferrous ammonium sulfate is a pale green crystalline compound which does not effloresce like ferrous sulfate.
Why the concentrated solution should not be heated for a long time?
The concentrated solution is stronger than other normal solution. … If they had been heated for a long time there may be chances to increase their strongness and make them highly concentrated which can be hardest to handle.
Why is ferrous ammonium sulphate solution not heated before titration?
In this titration heating of ferrous ammonium sulphate solution is not required because reaction rate is very high even at room temperature. Also, at high temperatures, ferrous ions may be oxidised to ferric ions by oxygen of air and error may be introduced in the experiment.
What is the purpose of adding concentrated sulphuric acid to the reaction mixture?
Heating the reaction mixture under reflux prevents the loss of volatile reactants and products. Concentrated sulfuric acid is used as a catalyst to speed up the rate at which the ester is formed (6).
Why do we add sulphuric acid before adding water for dissolving ferrous sulphate?
Fe2+ and Al3+ ions undergo hydrolysis, therefore, while preparing aqueous solutions of ferrous sulphate and aluminium sulphate in water, 2-3 mL dilute sulphuric acid is added to prevent the hydrolysis of these salts.
How does Sulphuric acid prevent hydrolysis?
How does acidification of aqueous copper sulphate solution prevent hydrolysis? – Quora. SO4 2- doesn’t hydrolyze appreciably as H2 SO4 is a fairly stronger acid than water. So on adding H+, due to common ion effect, the equilibrium of the Cu2+ hydrolysis is shifted to the left. In other words, hydrolysis decreases.
Why was sulfuric acid added to the iron sample before titrating?
Sulfuric acid is used because it is stable towards oxidation; whereas, for example, hydrochloric acid would be oxidized to chlorine by permanganate.
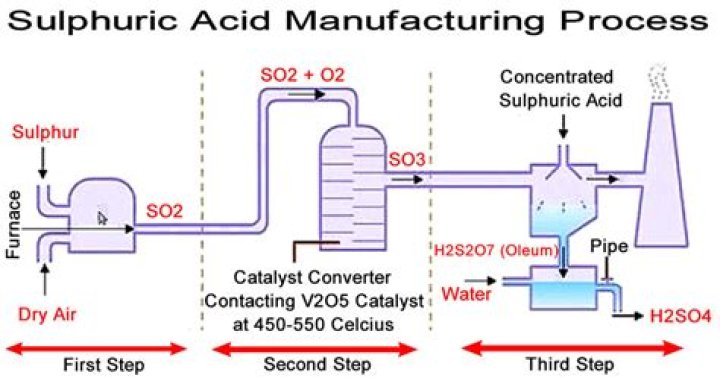
What is Sulphuric acid prepared for?
Sulfuric acid is prepared industrially by the reaction of water with sulfur trioxide (see sulfur oxide), which in turn is made by chemical combination of sulfur dioxide and oxygen either by the contact process or the chamber process. … Sulfuric acid is commonly supplied at concentrations of 78, 93, or 98 percent.
What is H2SO4 made up of?
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formula H2SO4.
Why do we add dilute H2SO4 in the titration between KMnO4 and oxalic acid?
The KMnO4 is an oxidizing agent that will oxidize oxalic acid. The H2SO4 ensures that all the oxalic acid remains as the acid to speed up the reaction.
Why did you heat solution oxalate and sulfuric acid mixture before titration with KMnO4?
Oxalic acid is heated before titration with KMnO4 solution because this reaction happens only in certain temperature. If not heated your light pink titrated solution turns brown soon & reaction takes longer times but overheating will cause decomposition of oxalic acid as CO2.
Why KMnO4 solution should not be added to rapidly during a titration?
The key reason for slow addition of KMnO4 is that it can do side reactions. Secondly, not all redox reactions are instantaneous. … The titration should be made in a solution very hot, in this way the reaction is faster.
Why is sulfuric acid stronger than phosphoric acid?
This is due to the fact that H2SO4 dissociates/deprotonates more in H2O than H3PO4 does. Stronger acids deprotonate almost completely in water, where weak acids dissociate incompletely in water, making H2SO4 a stronger acid, as the first Hydrogen atom dissociates completely, and the second does partially.
Why is a mixture of sulphuric acid and phosphoric acid used in the determination of Fe?
Usually H2SO4 and H3PO4 are added the sulfuric is the main source of H3O+ the phosphoric acid is added to complex Fe+3. By lowering the concentration of ferric ion the change in redox potential is increased making the endpoint easier to see or measure electronically.
What is the role of orthophosphoric acid?
phosphoric acid, also called orthophosphoric acid, (H3PO4), the most important oxygen acid of phosphorus, used to make phosphate salts for fertilizers. It is also used in dental cements, in the preparation of albumin derivatives, and in the sugar and textile industries.

