What is a balanced net ionic equation
The net ionic equation is the chemical equation that shows only those elements, compounds, and ions that are directly involved in the chemical reaction. … As a general rule, if you balance the molecular equation
How do you write a balanced net ionic equation?
- Write the balanced molecular equation.
- Write the state (s, l, g, aq) for each substance.
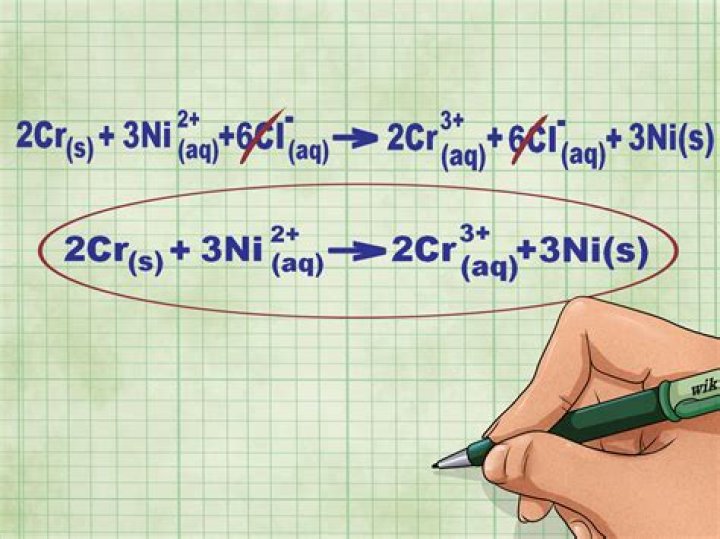
- Split strong electrolytes into ions (the complete ionic equation).
- Cross out the spectator ions on both sides of complete ionic equation.
What is the difference between a balanced equation and a net ionic equation?
The key difference between balanced equation and net ionic equation is that balanced equation shows all reactions that took place in a system together whereas net ionic equation only shows the net reaction that occurred after the completion of that particular reaction.
What is a balanced ionic equation?
A balanced ionic equation shows the reacting ions in a chemical reaction. These equations can be used to represent what happens in precipitation reactions .What is the difference between ionic and net ionic equations?
A net ionic equation shows only the chemical species that are involved in a reaction, while a complete ionic equation also includes the spectator ions.
Do you have to balance net ionic equations?
Net ionic equations must be balanced by both mass and charge. Balancing by mass means ensuring that there are equal masses of each element on the product and reactant sides. Balancing by charge means making sure that the overall charge is the same on both sides of the equation.
What is ionic and net ionic equations?
Complete ionic equation is a chemical equation that explains the chemical reaction, clearly indicating the ionic species present in a solution. The net ionic equation is a chemical equation which gives the ions that are participated in the formation of the final product.
What is a net reaction?
The net ionic equation is a chemical equation for a reaction that lists only those species participating in the reaction. The net ionic equation is commonly used in acid-base neutralization reactions, double displacement reactions, and redox reactions.Why do we write net ionic equations?
Net Ionic Equations Are Important The reason to write a chemical equation is to express what we believe is actually happening in a chemical reaction. … As such, they dissociate completely into their ions in solution, and although we might write “HCl” we really mean “H+ + Cl–”.
What is the difference between a chemical equation and a balanced chemical equation?An unbalanced chemical equation has an unequal number of atoms of one or more elements in the reaction. In a balanced chemical equation the sum of masses of the reactants is equal to the sum of the masses of the products. … Here, number of Zn, H, S and O are same in both reactants’ and products’ side.
Article first time published onCan a complete ionic equation and net ionic equation be the same?
As you know, spectator ions are ions that are present on both sides of the equation. In your case, the complete ionic equation and the net ionic equation will be one and the same, since no ions can be found on the reactants’ and on the products’ side.
What is a net ionic equation quizlet?
Net ionic equations are equations that show only the soluble, strong electrolytes reacting (these are represented as ions) and omit the spectator ions, which go through the reaction unchanged. … Ionic compounds are between metals and nonmetals or between metals and polyatomic ions.
What are net ions?
DEFINITIONS: spectator ion: an ion that is present in a solution in which a reaction takes place, but is not changed during the reaction process. net ionic equation: a chemical equation in which only those ions undergoing chemical changes during the course of the reaction are represented. Net Ionic Equations.
What is an ionic equation explain with an example?
An ionic equation is a chemical equation where the electrolytes in aqueous solution are written as dissociated ions. Example: 1) Sodium chloride(aq) + silver nitrate(aq) → silver chloride(s) + sodium nitrate(aq) >>Ag+(aq) + Cl-(aq) → AgCl(s) 2) Sodium(s) + hydrochloric acid(aq) -> sodium chloride(aq) + hydrogen(g).
What is the difference between skeletal and balanced equation?
In a balanced chemical reaction, the number of atoms in each element of a reactant side should be equal to the number of atoms of that element on the product side. In a skeletal equation, the number of atoms on the reactant side is not equal to the number of atoms on the product side.
Why must a chemical equation be balanced?
Chemical reactions are represented by chemical equations. All chemical equations must be balanced because matter cannot be created or destroyed in chemical reactions. Antoine Lavoisier did careful experiments to discover the law of conservation of mass in chemical reactions.
Is there a net ionic equation if there is no precipitate?
Precipitation reactions are usually represented solely by net ionic equations. If all products are aqueous, a net ionic equation cannot be written because all ions are canceled out as spectator ions. Therefore, no precipitation reaction occurs.
What does the net ionic equation of a precipitation reaction contain?
Subscripts are not needed to describe the state of the matter, because all ions are in aqueous solution. A net ionic equation is one in which spectator ions are removed. Spectator ions are present in solution but do not participate in the actual precipitation reaction.
Is NO3 always a spectator ion?
They are spectator ions because they did not take part in the chemical reaction. … The sodium ion has remained a spectator ion and the nitrate ion, NO3-, is also a spectator ion.
What information does a net ionic equation give that the complete ionic equation does not show?
Answer: The net ionic equation shows the chemical species involve in chemical reaction while the complete ionic equation also involve the spectator ions. Explanation: The net ionic equation shows the chemical species involve in chemical reaction while the complete ionic equation also involve the spectator ions.
How do you write a balanced equation for a neutralization reaction?
- acid + base → water + salt.
- HCl(aq) + KOH(aq) → H 2O(ℓ) + KCl(aq)
- 2HCl(aq) + Mg(OH) 2(aq) → 2H 2O(ℓ) + MgCl 2(aq)
- 3HCl(aq) + Fe(OH) 3(s) → 3H 2O(ℓ) + FeCl 3(aq)
- HCl(aq) + NaOH(aq) → H 2O(ℓ) + NaCl(aq)
- H +(aq) + Cl −(aq) + Na +(aq) + OH −(aq) → H 2O(ℓ) + Na +(aq) + Cl −(aq)
What is the net ionic equation for the reaction of any strong acid with a soluble strong hydroxide base such as NaOH group of answer choices?
Strong acids and strong bases are considered strong electrolytes and will dissociate completely. This means that we will split them apart in the net ionic equation. HNO3 + NaOH = NaNO3 + H2O is a neutralization reaction (also a double displacement reaction).

