What is the maximum concentration of ethanol in water by simple distillation and fractional distillation Why
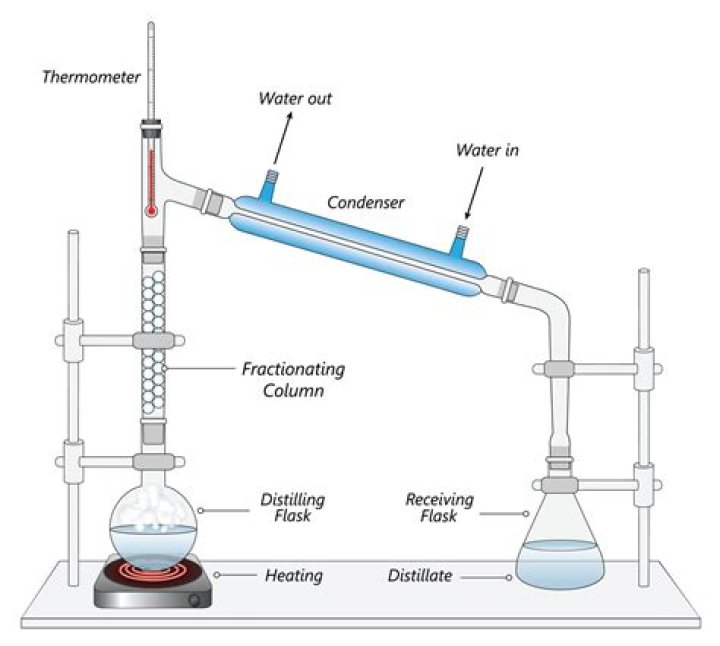
The liquid collected by condensing the vapor from the top of the fractionating column cannot be pure ethanol. The best you can produce by simple fractional distillation is 95.6% ethanol.
What is the maximum concentration of ethanol in water by fractional distillation?
The highest concentration of ethanol by volume that can be reached using fractional distillation is 96% which yields 192 proof solution.
What is the highest purity of ethanol than can be made by distillation?
95% Ethanol (190 Proof) 95% ethanol is the highest proof of alcohol achieved through only distillation without extraordinary processing techniques.
Why is fractional distillation better for ethanol and water?
Fractional distillation is a method for separating a liquid from a mixture of two or more liquids. For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points.Why is fractional distillation more efficient than simple distillation?
Fractional distillation leads to a better separation than simple distillation because the glass beads in the fractionating column provide “theoretical plates” on which the vapors can condense and then re-evaporate, and re-condense, essentially distilling the compound many times over.
What is distillation ethanol?
Distillation means taking the fermented ethanol and water mixture and adding heat to separate them — typically in a still. … Since ethanol evaporates faster than water, the ethanol rises through a tube, collects and condenses into another container. The water is left behind.
What is the concentration of the ethanol water mixture in the azeotrope?
A well-known example of a positive azeotrope is 95.63% ethanol and 4.37% water (by mass), which boils at 78.2 °C. Ethanol boils at 78.4 °C, water boils at 100 °C, but the azeotrope boils at 78.2 °C, which is lower than either of its constituents.
Is simple distillation vs fractional distillation?
Main Difference – Fractional Distillation vs Simple Distillation. … The main difference between fractional distillation and simple distillation is that simple distillation separate liquids with boiling point gaps of at least 50 degrees whereas fractional distillation separates liquids with closer boiling points.Which is more volatile water or ethanol?
Correct Option: A. Drinking alcohol (ethanol) and a lot of other simple alcohols are more volatile than water because they are less polar. Thus, alcohol has higher vapor pressure and a lower boiling point than water; therefore, it evaporates more quickly.
Is ethanol and water a homogeneous mixture?The mixture of ethanol and water is a type of homogeneous mixture. This is because ethanol is soluble in water and hence it is not possible to distinguish the ethanal and water from the solution.
Article first time published onCan you get 100 ethanol?
Short answer, it isn’t. Since 100% ethanol is so hydrophilic it pulls water straight out of the atmosphere to mix with you won’t get 100%. Distillation only gets you up to ~95% after which you’ll you can use a desiccant to remove the water.
Is 200 proof ethanol the same as 100?
“200 proof” means 100% ethyl alcohol by volume. “190 proof” means 95% Alcohol by volume, the remainder 5% comprised of water.
Can you drink 100 percent ethanol?
You can get alcohol poisoning from any alcoholic beverage, including beer and wine. Pure ethanol, such as Everclear, will dry out your mouth and burn the throat, but as long as you just drink a little bit, you should be fine. Intoxicated, maybe, but fine.
Is simple distillation effective in separating ethanol from its aqueous mixture?
Ethanol is perhaps the most often-cited example; a mixture of 95.6 percent ethanol and 4.4 percent water will actually boil at a lower temperature than either ethanol or water. Consequently, simple distillation cannot change this mixture’s composition.
Which distillation process is more efficient?
The fractional distillation is more efficient, and is suited for mixtures of volatile liquids. The closer the difference in the boiling points, the more demanding the distillation.
What are the advantages and disadvantages of simple distillation vs fractional distillation?
Simple distillationAdvantagessimpler setup than fractional faster distillation times consumes less energy than fractional distillationDisadvantagesrequires the liquids to have large boiling point differences (>70oC) gives poorer separation than fractional distillation only works well with relatively pure liquids
Does water and ethanol have an azeotrope?
The most common example is the azeotrope between water and ethanol (grain alcohol). Water boils at 100 ºC and ethanol boils at 78.3 ºC. The mixture will boil at 78.2 ºC and have a composition of 95% ethanol and 5% water by volume. This is a binary azeotrope because it involves two components.
Why the ethanol water mixture is an azeotrope?
In the example of the water:ethanol azeotrope, the water is able to increase the amount of hydrogen bonding between ethanol molecules by increasing the density of hydrogen bond donors and acceptors per unit volume.
What is minimum and maximum boiling azeotrope?
When the non-ideal binary solution shows the negative deviation, it is known as a maximum boiling azeotrope. When the non-ideal binary solution shows a positive deviation, it is known as a minimum boiling azeotrope.
What happens if you mix ethanol and water?
When you mix the rubbing alcohol with water, the latter’s molecules make hydrogen bonds with the water molecules. The alcohol dissolves in the water to form a homogenous solution, so you cannot distinguish the alcohol and the water anymore.
Does ethanol dissolve in water?
Methanol, ethanol, n-propyl alcohol, isopropyl alcohol, and t-butyl alcohol are all miscible with water.
Why can water be separated from inky water by simple distillation?
Pure water can be separated from inky water by simple distillation because water evaporates leaving the ink particles behind. The ink particles does not evaporate.
Which is more dense water or ethanol?
Water is more dense than alcohol or oil because its molecules can pack closely together, which means that it has more mass in the same volume than either alcohol or oil.
Is ethanol volatile in water?
Ethanol is a clear, colourless liquid with a characteristic pleasant odour and burning taste. … Ethanol is used to dissolve other chemical substances and mixes readily with water and many organic liquids. Ethanol is considered a volatile organic compound by the National Pollutant Inventory.
Which is more volatile methanol or ethanol?
The volatility of a liquid depends on the intermolecular forces of attraction between the particles of the liquid. … Low boiling point liquids, such as ethoxyethane or ethanal, are volatile. Ethanol, b.p. 79ºC is more volatile than water, b.p.100ºC, whereas methanol, b.p. 56ºC, is more volatile than ethanol.
Why does the temperature rise abruptly in the fractional distillation and gradually in the simple distillation?
Answer: The head temperature rises as the vapors of lower-boiling compound fill the distillation head. The temperature drops because the lower-boiling compound finishes distilling before vapors of the higher-boiling compound can fill the distillation head, which then cause the head temperature to rise.
How do you separate ethanol and water?
To separate a mixture of alcohol (ethanol) and water, you can use a process known as fractional distillation. This technique relies on the fact that the compounds in the mixture have different boiling points.
Is ethanol 20% a homogeneous mixture?
Yes, absolute ethanol is a compound confirmed. So it is a homogeneous Yeah.
Is ethanol solution homogeneous?
The ethanol molecules rapidly disperse and the solution becomes homogeneous. The entropy of the ethanol-water solution is higher than the of either separately that is: there are more distinguishable arrangements of the molecules when they are mixed, than when they are separate.
Is ethyl alcohol homogeneous or heterogeneous?
Ethanol and water, after mixing, can very easily become a homogeneous mixture, because the two liquids are miscible, soluble in all proportions.
Can you drink 200 proof alcohol?
What will happen if I drink 200 proof ethanol? Well, other than the fact that it’ll probably make you hork if you try to drink it straight… you’ll get about twice as drunk as you would from consuming the same amount of 100 proof ethanol (and slightly more than twice as drunk as you would from 80 proof).